Producción de biomasa de la microalga Chlorella sp. en condiciones fotoautotróficas y mixotróficas con yogurt semidescremado
DOI:
https://doi.org/10.70099/BJ/2026.03.02.6Palabras clave:
Chlorella sp, Mixotrofía, Valorización de residuos lácteos,, Biomasa microalgal, Cinética de crecimiento, BiorrefineríaResumen
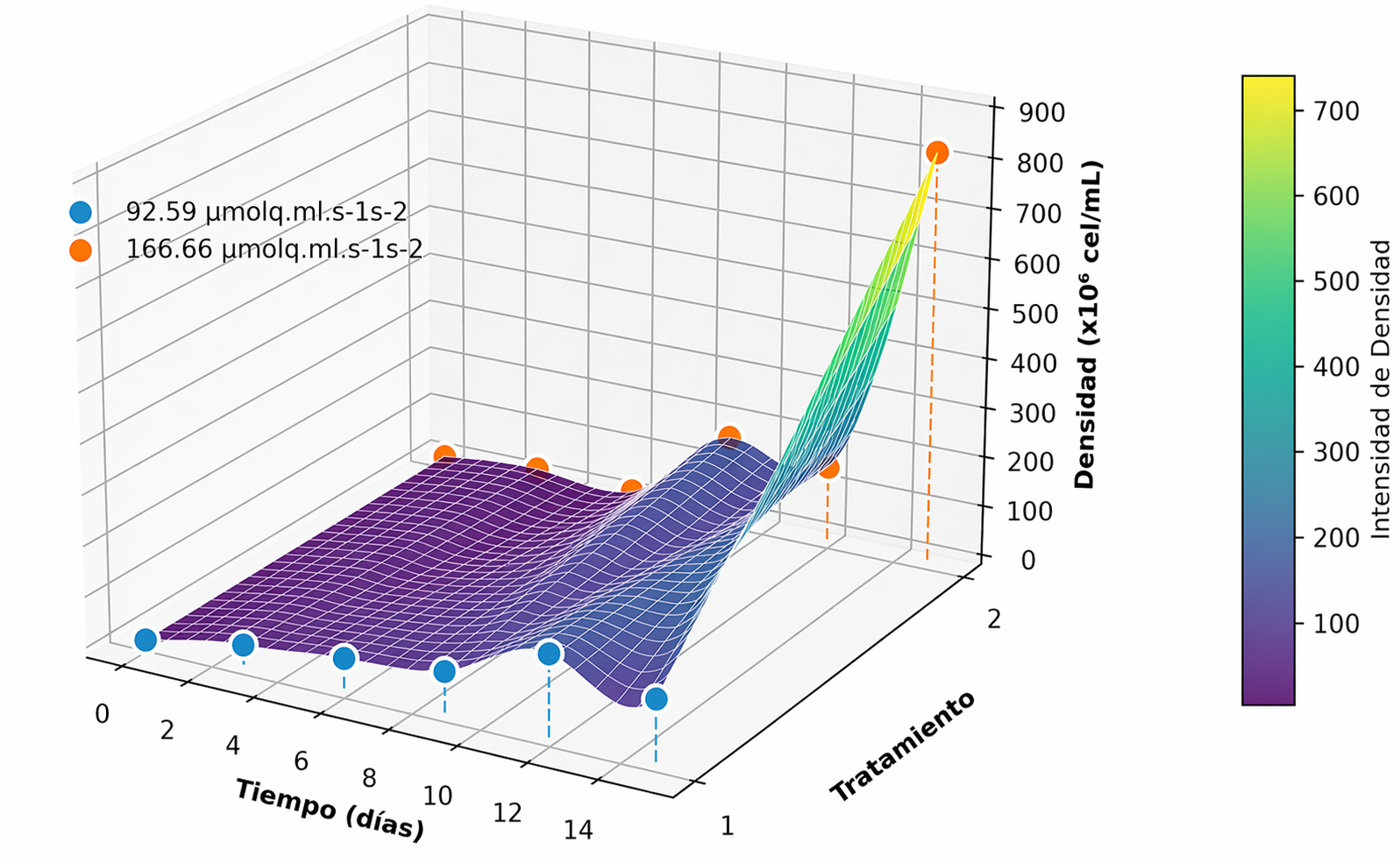
Se aisló la microalga Chlorella sp . a partir de un efluente de una industria láctea previamente sometida a tratabilidad biológica mediante un consorcio microbiano, considerando el aprovechamiento de residuos como alternativa para producir biomasa microalgal; el objetivo fue evaluar el crecimiento, la producción de pigmentos y la calidad bromatológica de la biomasa bajo condiciones autotróficas y mixotróficas, empleando fertilizante comercial Nitrofoska y yogurt natural semidescremado como fuentes de nutrientes. En cultivo autotrófico se procesan como condición óptima una concentración de 3 mL·L⁻¹ de fertilizante y una intensidad luminosa de 166.66 µmol·m⁻²·s⁻¹, con una densidad celular máxima de 26×10⁶ células y mayor producción de pigmentos liposolubles. En condiciones mixotróficas, el yogurt al 5 % indujo el mayor crecimiento de microalgas, alcanzando una densidad celular de 47.3×10⁶ (p = 0.0005), superior a las concentraciones de 10 % y 20 %. El análisis proximal mostró composiciones similares entre ambos cultivos, con contenidos proteicos de 38.3 % en mixotrofía y 34.9 % en autotrofía, el análisis mineral evidenció mayor acumulación de Ca y Na en cultivo mixotrófico, mientras que Fe, K, S, Pb y Cu aumentaron en condiciones autotróficas. El escalamiento mediante un sistema discontinuo alimentado permitió obtener productividades de biomasa de 3.16 g·L⁻¹ en cultivo mixotrófico y 0.81 g·L⁻¹ en autotrófico, sin diferencias relevantes en el costo de producción (0.04 y 0.03 ctvs·g⁻¹, respectivamente). En conclusión, el uso de derivados lácteos favorece la formación de consorcios de microalga-levadura, incrementa la productividad y constituye una alternativa viable para la producción de biomasa microalgal de bajo costo y proyección industrial.
Citas
1. Oslan SNH, Shoparwe NF, Yusoff AH, et al. A Review on Haematococcus pluvialis Bioprocess Optimi-zation of Green and Red Stage Culture Conditions for the Production of Natural Astaxanthin. Biomole-cules. 2021;11(2):256. doi:10.3390/biom11020256
2. Salbitani G, Carfagna S. Ammonium Utilization in Microalgae: A Sustainable Method for Wastewater Treatment. Sustainability. 2021;13(2):956. doi:10.3390/su13020956
3. Pereira L, Cotas J, Valado A. Antioxidants from microalgae and their potential impact on human well-being. Explor Drug Sci. 2024;2(3):292-321. doi:10.37349/eds.2024.00048
4. Capeletti L, Mossi A, Treichel H. EXTRACTION OF BIOPRODUCTS FROM MICROALGAE: A REVIEW OF METHODOLOGIES AND SCALE-UP STRATEGIES. Rev Multidiscip Nordeste Min. 2025;19:1-32. doi:10.61164/sgdfdc52
5. Paterson S, Gómez-Cortés P, de la Fuente MA, Hernández-Ledesma B. Bioactivity and Digestibility of Microalgae Tetraselmis sp. and Nannochloropsis sp. as Basis of Their Potential as Novel Functional Foods. Nutrients. 2023;15(2):477. doi:10.3390/nu15020477
6. Escobedo MJ, Calderón AC, Escobedo MJ, Calderón AC. Biomasa microalgal con alto potencial para la producción de biocombustibles. Sci Agropecu. 2021;12(2):265-282. doi:10.17268/sci.agropecu.2021.030
7. Szotkowski M, Holub J, Šimanský S, et al. Bioreactor Co-Cultivation of High Lipid and Carotenoid Pro-ducing Yeast Rhodotorula kratochvilovae and Several Microalgae under Stress. Microorganisms. 2021;9(6):1160. doi:10.3390/microorganisms9061160
8. Je S, Yamaoka Y. Biotechnological Approaches for Biomass and Lipid Production Using Microalgae Chlorella and Its Future Perspectives. 2022;32(11):1357-1372. doi:10.4014/jmb.2209.09012
9. González LM, Bashan LE. Toward the Enhancement of Microalgal Metabolite Production through Mi-croalgae–Bacteria Consortia. Biology. 2021;10(4):282. doi:10.3390/biology10040282
10. López Arias T, Galeano E, Santos M, Rodríguez Bonet S, Benitez A. Cinética de crecimiento y producti-vidad lipídica del alga Tetradesmus wisconsinensis S. obtenida en un cuerpo de agua de Paraguay. Inves-tig Agrar. 2016;18:22-29. doi:10.18004/investig.agrar.2016.junio.22-29
11. Sartory DP. The determination of algal chlorophyllous pigments by high performance liquid chromatog-raphy and spectrophotometry. Water Res. 1985;19(5):605-610. doi:10.1016/0043-1354(85)90066-1
12. Montoya C, Guzmán Duque FL, Quintero Díaz JC. Biomass and lipid production by the native green mi-croalgae Chlorella sorokiniana in response to nutrients, light intensity, and carbon dioxide: experimental and modeling approach. Front Bioeng Biotechnol. 2023;11. doi:10.3389/fbioe.2023.1149762
13. Michael A, Mtaki K. Optimizing Growth Conditions and Biomass Accumulation for Chlorella vulgaris of the Western Indian Ocean, Tanzania. Int J Biol Chem Sci. 2024;18(6):2066-2077. doi:10.4314/ijbcs.v18i6.1
14. Yaakob MA, Mohamed RMSR, Al-Gheethi A, Gokare RA, Ambati RR. Influence of Nitrogen and Phosphorus on Microalgal Growth, Biomass, Lipid, and Fatty Acid Production: An Overview. Cells. 2021;10(2). doi:10.3390/cells10020393
15. Takahashi M, Karitani Y, Yamada R, Matsumoto T, Ogino H. Co-utilization of microalgae and hetero-trophic microorganisms improves wastewater treatment efficiency. Appl Microbiol Biotechnol. 2024;108(1):468. doi:10.1007/s00253-024-13309-w
16. Mazzocchi E, Usai G, Agostino V, et al. Combination of Exhaust Gas Fermentation Effluent and Dairy Wastewater for Microalgae Production: Effect on Growth and FAME Composition of Chlorella sorokin-iana. Microorganisms. 2025;13(5):961. doi:10.3390/microorganisms13050961
17. Thepsuthammarat K, Reungsang A, Plangklang P. Microalga Coelastrella sp. Cultivation on Unhydro-lyzed Molasses-Based Medium towards the Optimization of Conditions for Growth and Biomass Produc-tion under Mixotrophic Cultivation. Molecules. 2023;28(8):3603. doi:10.3390/molecules28083603
18. Mountourakis F, Papazi A, Maragkoudakis A, Stamatis N, Kotzabasis K. Evidencia de adaptación fisioló-gica de Chlorella vulgaris bajo salinidad extrema: nuevos conocimientos sobre una posible estrategia de halotolerancia. Environ Exp Bot. 2023;216:105543. doi:10.1016/j.envexpbot.2023.105543
19. Yamaoka Y, Petroutsos D, Je S, Yamano T, Li-Beisson Y. Luz, CO2 y almacenamiento de carbono en microalgas. Curr Opin Plant Biol. 2025;84:102696. doi:10.1016/j.pbi.2025.102696
20. Gao P, Guo L, Gao M, Zhao Y, Jin C, She Z. Regulation of carbon source metabolism in mixotrophic microalgae cultivation in response to light intensity variation. J Environ Manage. 2022;302:114095. doi:10.1016/j.jenvman.2021.114095
21. Farooq W. Maximizing Energy Content and CO2 Bio-fixation Efficiency of an Indigenous Isolated Mi-croalga Parachlorella kessleri HY-6 Through Nutrient Optimization and Water Recycling During Culti-vation. Front Bioeng Biotechnol. 2022;9. doi:10.3389/fbioe.2021.804608
22. Mazzocchi E, Usai G, Agostino V, et al. Combination of Exhaust Gas Fermentation Effluent and Dairy Wastewater for Microalgae Production: Effect on Growth and FAME Composition of Chlorella sorokin-iana. Microorganisms. 2025;13(5). doi:10.3390/microorganisms13050961
23. Chai S, Shi J, Huang T, et al. Characterization of Chlorella sorokiniana growth properties in monosaccha-ride-supplemented batch culture. PLOS ONE. 2018;13(7):e0199873. doi:10.1371/journal.pone.0199873
24. Amaro HM, Salgado EM, Nunes OC, Pires JCM, Esteves AF. Microalgae systems - environmental agents for wastewater treatment and further potential biomass valorisation. J Environ Manage. 2023;337:117678. doi:10.1016/j.jenvman.2023.117678
25. Gómez L, Tormos-Cedeño L, Ortega-Díaz Y, Gómez-Luna L, Tormos-Cedeño L, Ortega-Díaz Y. Culti-vo y aplicaciones de Chlorella vulgaris: principales tendencias y potencialidades en la agricultura. Tecnol Quím. 2022;42(1):70-93.
26. González JF, Cuello TB, Calderón AJ, Calderón M, González J, Carmona D. Cultivation of Autochtho-nous Microalgae for Biomass Feedstock: Growth Curves and Biomass Characterization for Their Use in Biorefinery Products. Energies. 2021;14(15):4567. doi:10.3390/en14154567
27. Pang N, Gu X, Chen S, Kirchhoff H, Lei H, Roje S. Exploiting mixotrophy for improving productivities of biomass and co-products of microalgae. Renew Sustain Energy Rev. 2019;112:450-460. doi:10.1016/j.rser.2019.06.001
28. Patel AK, Joun J, Sim SJ. A sustainable mixotrophic microalgae cultivation from dairy wastes for carbon credit, bioremediation and lucrative biofuels. Bioresour Technol. 2020;313:123681. doi:10.1016/j.biortech.2020.123681
29. Salinas M, Calatrava-Arrizabalaga P, Ciardi M, Villaró-Cos S, Lafarga T. Development and reutilisation of a fertiliser-based culture medium for the commercial production of Chlorella sorokiniana. Sci Rep. 2025;15(1):23891. doi:10.1038/s41598-025-08320-8
30. Arashiro LT, Josa I, Ferrer I, Van Hulle SWH, Rousseau DPL, Garfí M. Life cycle assessment of micro-algae systems for wastewater treatment and bioproducts recovery: Natural pigments, biofertilizer and bio-gas. Sci Total Environ. 2022;847:157615. doi:10.1016/j.scitotenv.2022.157615
31. Abreu AP, Morais RC, Teixeira JA, Nunes J. A comparison between microalgal autotrophic growth and metabolite accumulation with heterotrophic, mixotrophic and photoheterotrophic cultivation modes. Renew Sustain Energy Rev. 2022;159:112247. doi:10.1016/j.rser.2022.112247
32. Yan X, Shan S, Li X, et al. Carbon and energy metabolism for the mixotrophic culture of Chlorella vul-garis using sodium acetate as a carbon source. Front Microbiol. 2024;15. doi:10.3389/fmicb.2024.1436264
33. Khoobkar Z, Delavari Amrei H. Efecto de las condiciones fototróficas, heterotróficas y mixotróficas sobre el crecimiento y la composición de Anabaena variabilis : Un enfoque de Energy Nexus. Energy Nexus. 2021;2:100010. doi:10.1016/j.nexus.2021.100010
34. Ge S, Qiu S, Tremblay D, Viner K, Champagne P, Jessop PG. Centrate wastewater treatment with Chlo-rella vulgaris: Simultaneous enhancement of nutrient removal, biomass and lipid production. Chem Eng J. 2018;342:310-320. doi:10.1016/j.cej.2018.02.058
35. Cai Y, Zhai L, Fang X, et al. Effects of C/N ratio on the growth and protein accumulation of heterotrophic Chlorella sp. in broken rice hydrolysate. Biotechnol Biofuels Bioprod. 2022;15(1):102. doi:10.1186/s13068-022-02204-z
36. Yun HS, Kim YS, Yoon HS. Effect of Different Cultivation Modes (Photoautotrophic, Mixotrophic, and Heterotrophic) on the Growth of Chlorella sp. and Biocompositions. Front Bioeng Biotechnol. 2021;9. doi:10.3389/fbioe.2021.774143
37. Xu Q, Hou G, Chen J, et al. Heterotrophically Ultrahigh-Cell-Density Cultivation of a High Pro-tein-Yielding Unicellular Alga Chlorella sp. With a Novel Nitrogen-Supply Strategy. Front Bioeng Bio-technol. 2021;9. doi:10.3389/fbioe.2021.774854
38. Hajri AK, Alsharif I, Albalawi MA, Alshareef SA, Albalawi RK, Jamoussi B. Utilizing Mixed Cultures of Microalgae to Up-Cycle and Remove Nutrients from Dairy Wastewater. Biology. 2024;13(8):591. doi:10.3390/biology13080591
39. Akhmedkhanova R, Dzhambulatov Z, Gadzhaeva Z, Shabanov G, Alieva S. The influence of Chlorella suspension on the quality of milk and its processing products. E3S Web Conf. 2020;222:02021. doi:10.1051/e3sconf/202022202021
40. Abate R, Oon YS, Oon YL, Bi Y. Microalgae-bacteria nexus for environmental remediation and renewa-ble energy resources: Advances, mechanisms and biotechnological applications. Heliyon. 2024;10(10). doi:10.1016/j.heliyon.2024.e31170
41. Abreu AP, Martins R, Nunes J. Emerging Applications of Chlorella sp. and Spirulina (Arthrospira) sp. Bioengineering. 2023;10(8):955. doi:10.3390/bioengineering10080955
42. Di Caprio F, Del Signore F, Capobianco L, Pagnanelli F, Altimari P. Feast–Famine in Cyclic Autotro-phy/Heterotrophy Doubles Microalgal Productivity while Controlling Bacterial Contamination. ACS Sus-tain Chem Eng. 2025;13(39):16448-16458. doi:10.1021/acssuschemeng.5c05990
43. Youssef AM, Gomaa M, Mohamed AKSH, El-Shanawany ARA. Enhancement of biomass productivity and biochemical composition of alkaliphilic microalgae by mixotrophic cultivation using cheese whey for biofuel production. Environ Sci Pollut Res. 2024;31(30):42875-42888. doi:10.1007/s11356-024-33877-8
44. Castillo OS, Torres-Badajoz SG, Núñez-Colín CA, et al. Producción de biodiésel a partir de microalgas: avances y perspectivas biotecnológicas. Hidrobiológica. 2017;27(3):337-352.
45. Braun JCA, Balbinot L, Beuter MA, Rempel A, Colla LM. Mixotrophic cultivation of microalgae using agro-industrial waste: Tolerance level, scale up, perspectives and future use of biomass. Algal Res. 2024;80:103554. doi:10.1016/j.algal.2024.103554
46. Feng L, Guo W, Guo J, et al. FIB-SEM analysis on three-dimensional structures of growing organelles in wild Chlorella sp. pyrenoidosa cells. Protoplasma. 2023;260(3):885-897. doi:10.1007/s00709-022-01821-7
47. Chen H, Zheng Y, Zhan J, He C, Wang Q. Comparative metabolic profiling of the lipid-producing green microalga Chlorella sp. reveals that nitrogen and carbon metabolic pathways contribute to lipid metabo-lism. Biotechnol Biofuels. 2017;10(1):153. doi:10.1186/s13068-017-0839-4
Publicado
Cómo citar
Licencia
Derechos de autor 2026 Ever Morales Avendaño, Geovanna Elizabeth Guevara Figueroa , Junior Jesús Vélez Espinoza , Pedro José Rachid Romero Saker (Author)

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
Authors who publish with BioNatura Journal agree to the following terms: Authors retain copyright and grant the BioNatura Institutional Publishing Consortium (BIPC) right of first publication with the work simultaneously licensed under a Creative Commons Attribution 4.0 International License (CC BY 4.0). This allows others to share the work with an acknowledgment of the work's authorship and initial publication in this journal.This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.




