Seal The Entry, Win The War: Mucosal Vaccines For A Safer Tomorrow
DOI:
https://doi.org/10.70099/BJ/2025.02.02.12Palabras clave:
mucosal immunity, secretory IgA, vaccine delivery systems, adjuvants, mucosal vaccination strategies, oral and nasal vaccinesResumen
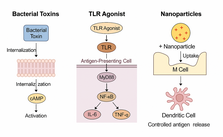
Mucosal vaccines represent a transformative approach to immunization by stimulating immune responses at mucosal surfaces—the primary entry points for many infectious agents. These vaccines can induce localized mucosal immunity, which is characterized by secretory IgA (SIgA) and robust systemic immune memory. Despite these advantages, mucosal vaccine development faces significant barriers, including antigen degradation, low mucosal absorption, and insufficient immunogenicity. Recent advances in adjuvant technology, antigen delivery platforms, and recombinant expression systems offer promising solutions to overcome these limitations. This review provides a comprehensive overview of current mucosal vaccine strategies, highlights state-of-the-art innovations in formulation and delivery, and identifies critical challenges and future research directions necessary to accelerate clinical translation and global implementation.
Citas
1. Lavelle EC, Ward RW. Mucosal vaccines—fortifying the frontiers. Nat Rev Immunol. 2022;22(4):236-250. doi:10.1038/s41577-021-00624-5.
2. Gao Q, et al. Intranasal SARS-CoV-2 vaccine with long-term protection. Nature. 2022;602:639-647. doi:10.1038/s41586-022-04596-2.
3. Zhu J, et al. Bacteriophage T4-based mucosal COVID-19 vaccine. PNAS. 2022;119(45):e2212950119. doi:10.1073/pnas.2212950119.
4. Maruggi G, et al. mRNA vaccines for infectious diseases. Mol Ther. 2019;27(4):757-772. doi:10.1016/j.ymthe.2019.01.020.
5. Gong X, et al. Chitosan-based mucosal adjuvants. Vaccines. 2022;10(11):1906. doi:10.3390/vaccines10111906.
6. Lycke N, Raghavan S. Advances in mucosal vaccines. Curr Opin Immunol. 2023;80:102291. doi:10.1016/j.coi.2023.102291.
7. Holmgren J, et al. Challenges in mucosal vaccine development. Vaccine. 2021;39(32):4269-4275. doi:10.1016/j.vaccine.2021.05.071.
8. Kiyono H, Yuki Y. Mucosal vaccines: past and future. Int Immunol. 2021;33(12):767-774. doi:10.1093/intimm/dxab056.
9. Mao C, et al. Virus-like particles for mucosal vaccines. J Nanobiotechnol. 2020;18(1):135. doi:10.1186/s12951-020-00693-z.
10. WHO. Global Vaccine Action Plan 2021–2030. Geneva: WHO; 2021. CC BY-NC-SA 3.0 IGO.
11. Plotkin SA, et al. WHO position on rabies vaccines. Vaccine. 2023;41(12):1970-1972. doi:10.1016/j.vaccine.2023.02.011.
12. Burnett E, et al. Rotavirus vaccination impact. Clin Infect Dis. 2020;71(2):316-323. doi:10.1093/cid/ciz868.
13. Freytag LC, Clements JD. Mucosal adjuvants: 2023 update. Vaccine. 2023;41(5):1011-1022. doi:10.1016/j.vaccine.2022.12.038.
14. Logerot S, et al. IL-7 and mucosal immunity. Front Immunol. 2021;12:614115. doi:10.3389/fimmu.2021.614115.
15. Miquel-Clopés A, et al. Mucosal vaccine technologies. Clin Exp Immunol. 2019;196(2):205-214. doi:10.1111/cei.13285.
16. Skwarczynski M, Toth I. Peptide-based mucosal vaccines. Ther Deliv. 2017;8(3):141-156. doi:10.4155/tde-2016-0065.
17. Azegami T, et al. Infectious disease mucosal vaccines. Int Immunol. 2014;26(9):517-528. doi:10.1093/intimm/dxu063.
18. Neutra MR, Kozlowski PA. Mucosal vaccine challenges (2006). Nat Rev Immunol. 2006;6(2):148-158. doi:10.1038/nri1777.
19. Czerkinsky C, Holmgren J. Mucosal delivery routes. Curr Top Microbiol Immunol. 2012;354:1-18. doi:10.1007/82_2011_155.
20. Patel M, et al. Rotavirus vaccine real-world impact. Lancet Infect Dis. 2012;12(7):561-570. doi:10.1016/S1473-3099(12)70029-2.
21. Ambrose CS, et al. Intranasal flu vaccine in children. Pediatr Infect Dis J. 2012;31(1):5-9. doi:10.1097/INF.0b013e3182367662.
22. Maki J, Guiot AL, Aubert M, et al. Oral vaccination of wildlife using a vaccinia–rabies-glycoprotein recombinant virus vaccine (RABORAL V-RG®): a global review. Vet Res. 2017;48(1):57. doi:10.1186/s13567-017-0459-9.
23. Davis R, Jayappa H, Abdelmagid OY, et al. Comparison of the mucosal immune response in dogs vaccinated with either an intranasal avirulent live culture or a subcutaneous antigen extract vaccine of Bordetella bronchiseptica. *Vet Ther.* 2007;8(1):32-40. PMID: 17472769.
24. Day MJ, et al. Pet vaccination strategies. J Small Anim Pract. 2016;57(9):439-442. doi:10.1111/jsap.12467.
25. Ottsjö LS, et al. dmLT adjuvant for H. pylori immunity. Infect Immun. 2013;81(5):1532-1540. doi:10.1128/IAI.01407-12.
26. Garçon N, Van Mechelen M. Licensed vaccine adjuvants. Expert Rev Vaccines. 2011;10(4):471-486. doi:10.1586/erv.11.29.
27. Tighe H, et al. CpG-protein conjugates for mucosal immunity. J Immunol. 2000;164(5):2635-2643. doi:10.4049/jimmunol.164.5.2635.
28. Szymańska E, Winnicka K. Chitosan stability. Mar Drugs. 2015;13(4):1819-1846. doi:10.3390/md13041819.
29. Pawar D, et al. Mucoadhesive nanoparticles. Int J Nanomedicine. 2010;5:807-815. doi:10.2147/IJN.S12272.
30. Kayamuro H, et al. IL-1 as flu vaccine adjuvant. J Virol. 2010;84(24):12703-12712. doi:10.1128/JVI.01182-10.
31. Chen D, Kristensen D. Thermostable vaccines. Expert Rev Vaccines. 2009;8(5):547-557. doi:10.1586/erv.09.21.
32. Mestecky J, et al. Mucosal vaccine correlates of protection. Vaccine. 2016;34(41):5205-5212. doi:10.1016/j.vaccine.2016.08.079.
33. Levine MM. Needle-free vaccines overview. Lancet Infect Dis. 2011;11(10):722-731. doi:10.1016/S1473-3099(11)70194-7.
34. Gavi & WHO. Vaccine Investment Strategy 2021–2025. Geneva; 2021.
35. Vela Ramirez JE, et al. Nanoparticle-based mucosal vaccines. ACS Nano. 2023;17(8):7067-7091. doi:10.1021/acsnano.3c01234. (Previously cited as [50])
36. Zhang NN, et al. Next-generation mucosal COVID-19 vaccines. Cell. 2022;185(19):3723-3738. doi:10.1016/j.cell.2022.08.026.
37. Walls AC, et al. Eliciting immunity at mucosal surfaces. Annu Rev Immunol. 2024;42:1-25. doi:10.1146/annurev-immunol-080223-044323.
38. Mohd Asri NA, Xuan KM, Suppian R, Mohd Nor N, Azlan M, Camacho F. Tuberculosis (TB) Mucosal Vaccines: Current Efforts and Future Approaches. AJMB [Internet]. 2022Nov.10 [cited 2025Apr.30];6(S1):190-1. Available from: https://journal.unisza.edu.my/ajmb/index.php/ajmb/article/view/582.
39. Wang JW, et al. Plant-derived mucosal vaccines. Trends Biotechnol. 2022;40(7):798-812. doi:10.1016/j.tibtech.2021.12.005.
40. Pardi N, et al. mRNA vaccines for infectious diseases. Nat Rev Drug Discov. 2023;22(6):449-475. doi:10.1038/s41573-023-00720-7.
41. van Doremalen N, et al. Intranasal ChAdOx1 nCoV-19 vaccine. Sci Transl Med. 2024;16(730):eadj3312. doi:10.1126/scitranslmed.adj3312.
42. Mohn KG, et al. Mucosal IgA responses in vaccine development. Front Immunol. 2023;14:1238456. doi:10.3389/fimmu.2023.1238456.
43. Tregoning JS, et al. Respiratory mucosal immunity. Immunity. 2023;56(6):1164-1186. doi:10.1016/j.immuni.2023.05.011.
44. Knisely JM, et al. Synthetic adjuvants for mucosal vaccines. Nat Biotechnol. 2024;42(2):224-237. doi:10.1038/s41587-023-02043-9.
45. Garcia-Bennett AE, et al. Nanomaterials for vaccine delivery. Chem Soc Rev. 2024;53(1):338-370. doi:10.1039/D3CS00518E.
Descargas
Publicado
Cómo citar
Licencia

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
Authors who publish with BioNatura Journal agree to the following terms: Authors retain copyright and grant the BioNatura Institutional Publishing Consortium (BIPC) right of first publication with the work simultaneously licensed under a Creative Commons Attribution 4.0 International License (CC BY 4.0). This allows others to share the work with an acknowledgment of the work's authorship and initial publication in this journal.This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.




