Development and Validation of a Phenomenological Model for Scalable Size-Exclusion Chromatography in TNF-Blocking Peptide Purification
DOI:
https://doi.org/10.70099/BJ/2025.02.02.2Palabras clave:
size exclusion chromatography, phenomenological model, TNF blocking agent, peptides, bioprocess modeling, in silico simulation, scale-upResumen
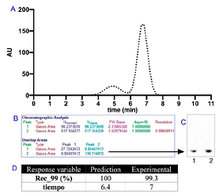
A recombinant peptide as a TNF-blocking agent was obtained using fermentation in E. coli. The strategy was based on the expression of a multimeric tandem protein comprising 15 repeated copies of the r-CBB288 peptide, and the monomeric unit was released with a hydroxylamine-assisted proteolysis step. At this point, size exclusion chromatography was used to separate the monomer unit from unreacted polymer forms. Therefore, the main theme of this study was to develop a phenomenological General Rate Model to describe the elution profile of SEC and to ensure the required purity levels for the mentioned peptide. In that sense, with the development of the phenomenological model, it will be possible to design different process scales of desired dimensions further. For such purpose, the reproducibility of the SEC was determined on a lab scale, and phenomenological coefficients were calculated from mathematical correlations and simulated in the Matlab software. For the validation of the mathematical model, a DoE (23 with central point) was performed, where the sample load, velocity, and bed height were assessed as independent variables, whereas 99% purity and operating time were response variables. As the main results, simulated profiles did not show statistical differences (residual ≤ 5%) concerning experimental profiles. This study introduces a validated phenomenological model for SEC applied to purifying a TNF-blocking peptide, integrating computational simulations with experimental validation, enabling reliable process scale-up for industrial bioprocess design. In conclusion, it was possible to develop a phenomenological model of the SEC to ensure the purity of the antiTNF recombinant prototype at different scales, which allows the designing of industrial processes at scales of desired dimensions.
Citas
1. Gu T. Mathematical Modeling and Scale-Up of Liquid Chromatography: With Application Examples: Springer; 2015 2015/04/06/. 214 p.
2. Maqsoudlou A, Mahoonak AS, Mora L, Mohebodini H, Toldra F, Ghorbani M. Peptide identification in alcalase hydrolysated pollen and comparison of its bioactivity with royal jelly. Food Research International. 2019;116:905-15.
3. Preparative Chromatography of Fine Chemicals and Pharmaceutical Agents. Oxford: Blackwell Science Publ; 2006 2006.
4. Harrison RG, Todd P, Rudge SR, Petrides DP. Bioseparations Science and Engineering: Oxford University Press; 2015 2015. 577 p.
5. Jagschies G, Sofer GK, Hagel L. Handbook of Process Chromatography: Development, Manufacturing, Validation and Economics: Elsevier; 2007 2007/12/08/. 383 p.
6. Zelic B, Nesek B. Mathematical Modeling of Size Exclusion Chromatography. Eng Life Sci. 2006;6(2):163-9.
7. Faraji N, Zhang Y, Ray AK. Impact of operating conditions on chromatographic column performance: experimental studies on adsorption of high-value minor whey proteins. AIMS Bioeng. 2017;4(2):223-38.
8. Chester TL. The combination of partition, size exclusion, and hydrodynamic models in chromatography, and application to bonded phases on porous supports. Journal of Chromatography A. 2020;1620:461011.
9. Li ZG, Gu YS, Gu TY. Mathematical modeling and scale-up of size-exclusion chromatography. Biochemical Engineering Journal. 1998;2(2):145-55.
10. Pavlov G, Hsu JT. Modelling the effect of temperature on the gel-filtration chromatographic protein separation. Computers & Chemical Engineering. 2018;112:304-15.
11. Guiochon G, Felinger A, Shirazi DGG. Fundamentals of Preparative and Nonlinear Chromatography: Elsevier; 2006 2006/02/10/. 991 p.
12. Reinglas J, Lorant Gonczi ZK, Bessissow T, Lakatos PL. Positioning of old and new biologicals and small molecules in the treatment of inflammatory bowel diseases. World Journal of Gastroenterology. 2018;24(32):3567.
13. Richmond V, M Michelini F, A Bueno C, E Alche L, A Ramirez J. Small molecules as anti-TNF drugs. Current Medicinal Chemistry. 2015;22(25):2920-42.
14. Li P, Zheng Y, Chen X. Drugs for Autoimmune Inflammatory Diseases: From Small Molecule Compounds to Anti-TNF Biologics. Frontiers in Pharmacology. 2017;8.
15. Kalliolias GD, Ivashkiv LB. TNF biology, pathogenic mechanisms and emerging therapeutic strategies. Nature Reviews Rheumatology. 2016;12(1):49-62.
16. Schägger H. Tricine–SDS-PAGE. Nature Protocols. 2006;1(1):16-22.
17. Gritti F, Guiochon G. Application of the general height equivalent to a theoretical plate equation to size exclusion chromatography. Study of the mass transfer of high-molecular-mass compounds in liquid chromatography. Anal Chem. 2007;79(8):3188-98.
18. Chester TL. The combination of partition, size exclusion, and hydrodynamic models in chromatography... J Chrom A. 2020;1620:461011.
19. Pavlov G, Hsu JT. Modelling the effect of temperature on gel-filtration chromatographic protein separation. Comput Chem Eng. 2018;112:304–15.
20 Faraji N, Zhang Y, Ray AK. Digital twins in chromatography: predictive modeling in bioseparation process optimization. AIMS Bioengineering. 2021;8(2):223-240.
Descargas
Publicado
Cómo citar
Número
Sección
Categorías
Licencia

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
Authors who publish with BioNatura Journal agree to the following terms: Authors retain copyright and grant the BioNatura Institutional Publishing Consortium (BIPC) right of first publication with the work simultaneously licensed under a Creative Commons Attribution 4.0 International License (CC BY 4.0). This allows others to share the work with an acknowledgment of the work's authorship and initial publication in this journal.This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.




