RNA Technologies in Cancer Care: Bridging Promise and Practice
DOI:
https://doi.org/10.70099/BJ/2025.02.02.15Keywords:
RNA-seq, non-coding RNAs, mRNA vaccines, precision oncology, RNA interference, biomarkers, nanodelivery, medical ethicsAbstract
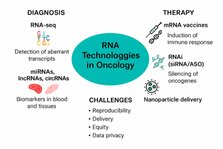
RNA technologies are rapidly reshaping cancer diagnostics and therapeutics, offering unprecedented promise alongside persistent challenges. This News and Views synthesizes evidence from meta-analyses and systematic reviews on RNA sequencing (RNA-seq), non-coding RNAs, and RNA-based therapies. We highlight advances in personalized diagnostics, mRNA vaccines, and RNA interference (RNAi), while addressing technical, clinical, and ethical barriers to clinical adoption. A balanced perspective is essential as the field advances toward precision oncology.
References
1. Byron SA, Van Keuren-Jensen KR, Engelthaler DM, Carpten JD, Craig DW. Translating RNA sequencing into clinical diagnostics: opportunities and challenges. Nat Rev Genet. 2016;17(5):257-269. doi:10.1038/nrg.2016.10.
2. Buzdin A, Sorokin M, Garazha A, et al. RNA sequencing for research and diagnostics in clinical oncology. Semin Cancer Biol. 2020;60:311-323. doi:10.1016/j.semcancer.2019.07.010.
3. Hong M, Tao S, Zhang L, et al. RNA sequencing: new technologies and applications in cancer research. J Hematol Oncol. 2020;13:166. doi:10.1186/s13045-020-01005-x.
4. Wang H, Meng Q, Qian J, et al. RNA-based diagnostic markers discovery and therapeutic targets development in cancer. Pharmacol Ther. 2022;239:108123. doi:10.1016/j.pharmthera.2022.108123.
5. Kristensen LS, Hansen TB, Venø MT, Kjems J. Circular RNAs in cancer: opportunities and challenges in the field. Oncogene. 2017;37:555-565. doi:10.1038/onc.2017.361.
6. Tortora F, La Civita E, Trivedi P, et al. Emerging RNA-Based Therapeutic and Diagnostic Options: Recent Advances and Future Challenges in Genitourinary Cancers. Int J Mol Sci. 2023;24(5):4601. doi:10.3390/ijms24054601.
7. Sempere LF, Azmi AS, Moore A. microRNA-based diagnostic and therapeutic applications in cancer medicine. Wiley Interdiscip Rev RNA. 2021;12(2):e1662. doi:10.1002/wrna.1662.
8. Liu C, Shi Q, Huang X, et al. mRNA-based cancer therapeutics. Nat Rev Cancer. 2023;23:241-258. doi:10.1038/s41568-023-00586-2.
9. Eralp Y. Application of mRNA Technology in Cancer Therapeutics. Vaccines (Basel). 2022;10(8):1262. doi:10.3390/vaccines10081262.
10. Attia MF, Kijanka G, Nguyen NL, et al. Advances and prospects of RNA delivery nanoplatforms for cancer therapy. Acta Pharm Sin B. 2024;15(3):456-472. doi:10.1016/j.apsb.2024.09.009.
11. Chu LH, Joly Y, Knoppers BM. Equity in access to gene and RNA therapies: mapping clinical trial inclusion. Sci Transl Med. 2023;15(719):eadd1902. doi:10.1126/scitranslmed.add1902.
12. Zavala VA, Kalinowski A, Sanchez HN, et al. Ethical considerations for sharing transcriptomic data in cancer research. EXCLI J. 2021;20:1331-1336. doi:10.17179/excli2021-3833.
Published
How to Cite
License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors who publish with BioNatura Journal agree to the following terms: Authors retain copyright and grant the BioNatura Institutional Publishing Consortium (BIPC) right of first publication with the work simultaneously licensed under a Creative Commons Attribution 4.0 International License (CC BY 4.0). This allows others to share the work with an acknowledgment of the work's authorship and initial publication in this journal.This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.




