Computational Comparison of the Binding Affinity of Selective and Nonselective NSAIDs to COX-2 Using Molecular Docking
DOI:
https://doi.org/10.70099/BJ/2025.02.02.3Keywords:
NSAIDs, active site, COX-2, Docking, selectivityAbstract
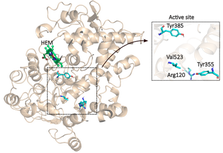
Cyclooxygenase-2 (COX-2) plays a key role in inflammation, making it a prime target for nonsteroidal anti-inflammatory drugs (NSAIDs). This study uses molecular docking to compare the binding affinities of four nonselective NSAIDs (aspirin, ibuprofen, diclofenac, naproxen) and three selective COX-2 inhibitors (celecoxib, rofecoxib, etoricoxib) to COX-2. Simulations with AutoDock4 and AutoDock Vina revealed distinct differences in binding profiles and selectivity. Selective COX-2 inhibitors exhibited stronger binding affinities, with etoricoxib achieving -11.22 kcal/mol (AutoDock4), driven by key hydrogen bonds and π interactions. Nonselective NSAIDs, such as diclofenac (-8.08 kcal/mol), showed moderate affinity but lacked specificity, targeting both COX isoforms and increasing gastrointestinal side effects. AutoDock4 provided detailed conformational analysis, while AutoDock Vina complemented with faster but less detailed results. This research highlights the structural interactions underlying NSAID efficacy and side effects, offering valuable insights for drug design. Selective inhibitors provide improved safety profiles for long-term use, while nonselective NSAIDs remain effective for short-term treatments. These findings emphasize the importance of computational tools in optimizing NSAID selectivity and efficacy, paving the way for developing safer anti-inflammatory therapies.
References
1. Martín-Vázquez E, Cobo-Vuilleumier N, López-Noriega L, Lorenzo PI, Gauthier BR. The PTGS2/COX2-PGE2 signaling cascade in inflammation: Pro or anti? A case study with type 1 diabetes mellitus. Int J Biol Sci. 2023;19(13):4157–65.
2. Salido M, Abásalo L, Bañares A. Revisión de los antiinflamatorios inhibidoresselectivos de la ciclooxigenasa-2. Inf Ter del Sist Nac Salud. 2001;25(2):46–52.
3. Rao PNP, Knaus EE. Evolution of Nonsteroidal Anti-Inflammatory Drugs (NSAIDs): Cyclooxygenase (COX) Inhibition and Beyond. J Pharm Pharm Sci [Internet]. 2008;11(2):81-110s. Available from: https://ejournals.library.ualberta.ca/index.php/JPPS/article/view/4128%0Ahttps://journals.library.ualberta.ca/jpps/index.php/JPPS/article/view/4128/3358
4. Ahmadi M, Bekeschus S, Weltmann KD, von Woedtke T, Wende K. Nonsteroidal anti-inflammatory drugs: recent advances in the use of synthetic COX-2 inhibitors. RSC Med Chem [Internet]. 2022;(February). Available from: 10.1039/d1md00280e
5. Bindu S, Mazumder S, Bandyopadhyay U. Nonsteroidal anti-inflammatory drugs (NSAIDs) and organ damage: A current perspective. Biochem Pharmacol. 2020;180(April).
6. Peura DA, Goldkind L. Balancing the gastrointestinal benefits and risks of nonselective NSAIDs. Arthritis Res Ther [Internet]. 2005;7(SUPPL. 4):7–13. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2833976/pdf/ar1793.pdf
7. Tomić M, Micov A, Pecikoza U, Stepanović-Petrović R. Clinical Uses of Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) and Potential Benefits of NSAIDs Modified-Release Preparations. Microsized Nanosized Carriers Nonsteroidal Anti-inflamm Drugs Formul Challenges Potential Benefits. 2017;1–29.
8. Muhammed MT, Aki-Yalcin E. Molecular Docking: Principles, Advances, and its Applications in Drug Discovery. Lett Drug Des Discov [Internet]. 2022;19:1–16. Available from: http://www.esisresearch.org/Uploads/Documents/esis2022muhammed(molecular-docking)lettdrugdesigndiscov.pdf
9. Novikov FN, Chilov GG. Molecular docking: theoretical background, practical applications and perspectives. Mendeleev Commun [Internet]. 2009;19(5):237–42. Available from: 10.1016/j.mencom.2009.09.001
10. Raval K, Ganatra T. Basics, types and applications of molecular docking: A review. IP Int J Compr Adv Pharmacol [Internet]. 2022;7(1):12–6. Available from: https://www.researchgate.net/profile/Keval-Raval-2/publication/359240621_Basics_types_and_applications_of_molecular_docking_A_review/links/6236ab9772d413197a33e328/Basics-types-and-applications-of-molecular-docking-A-review.pdf
11. Torrez G, Enrique R. Acomplamiento molecular: criterios prácticos para la selección de ligandos biológicamente activos e identificación de nuevos blancos terapéuticos. Rev Con-Ciencia. 2019;2:1–18.
12. Moussa N, Hassan A, Gharaghani S. Pharmacophore model, docking, QSAR, and molecular dynamics simulation studies of substituted cyclic imides and herbal medicines as COX-2 inhibitors. Heliyon [Internet]. 2021;7(4):e06605. Available from: https://doi.org/10.1016/j.heliyon.2021.e06605
13. Jara M, Jaramillo L, Matamoros JM. F recuencia de automedicación de AINES y analgésicos - antipiréticos y características que los rodean, en hogares de la parroquia San Blas de la ciudad de Cuenca en el año 2011 [Internet]. Universidad De Cuenca Facultad De Ciencias Médicas Escuela De Medicina. Universidad De Cuenc; 2011. Available from: http://dspace.ucuenca.edu.ec/bitstream/123456789/3466/1/MED95.pdf
14. Encalada C, Ortega J, Carlos Valencia. Prevalencia Y Factores Asociados a La Automedicación Con Aines En Adultos Mayores En Las Parroquias Urbanas De Cuenca [Internet]. Universidad de Cuenca. Universidad de Cuenca; 2015. Available from: http://dspace.ucuenca.edu.ec/bitstream/123456789/22494/1/tesis.pdf
15. Hanwell M, Curtis D, Lonie D, Vandermeersh T, Zurek E, Hutchison G. Avogadro: An advanced semantic chemical editor, visualization, and analysis platform. J Cheminform. 2012;
16. Morris G, Huey R, Lindstrom W, Sanner M, Belew R, Goodsell D, et al. Autodock4 and AutoDockTools4: automated docking with selective receptor flexiblity. Computational Chemistry 2009; 2009. p. 2785–91.
17. Eberhardt J, Santos-Martins D, Tillack A., Forli S. AutoDock Vina 1.2.0: New Docking Methods, Expanded Force Field, and Python Bindings. Journal of Chemical Information and Modeling; 2021.
18. RStudio Team. RStudio: Integrated Development for R. RStudio, PBC. 2020.
19. Schrödinger L, DeLano W. PyMOL. 2020.
20. BIOVIA DS. BIOVIA Discovery Studio [Software]. 2021.
21. Bello-Vargas E, Leyva-Peralta MA, Gómez-Sandoval Z, Ordóñez M, Razo-Hernández RS. A Computational Method for the Binding Mode Prediction of COX-1 and COX-2 Inhibitors: Analyzing the Union of Coxibs, Oxicams, Propionic and Acetic Acids. Pharmaceuticals. 2023;16(12).
22. Chaudhary N, Aparoy P. Deciphering the mechanism behind the varied binding activities of COXIBs through Molecular Dynamic Simulations, MM-PBSA binding energy calculations and per-residue energy decomposition studies. J Biomol Struct Dyn. 2017;35(4):868–82.
23. Ibrahim MM, Elsaman T, Al-Nour MY. Synthesis, Anti-Inflammatory Activity, and In Silico Study of Novel Diclofenac and Isatin Conjugates. Int J Med Chem. 2018;2018:1–11.
24. Meneses L, Cuesta S. Determinación Computacional de la Afinidad y Eficiencia de Enlace de Antinflamatorios No Esteroideos Inhibidores de la Ciclooxigenasa-2. Rev Ecuat Med Cienc Biol. 2015;36(1–2):17–25.
25. Sahu MK, Nayak AK, Hailemeskel B, Eyupoglu OE. Exploring Recent Updates on Molecular Docking: Types, Method, Application, Limitation & Future Prospects. Int J Pharm Res Allied Sci. 2024;13(2):24–40.
26. Tobi D, Bahar I. Optimal design of protein docking potentials: Efficiency and limitations. Proteins Struct Funct Genet. 2006;62(4):970–81
Published
How to Cite
Issue
Section
Categories
License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors who publish with BioNatura Journal agree to the following terms: Authors retain copyright and grant the BioNatura Institutional Publishing Consortium (BIPC) right of first publication with the work simultaneously licensed under a Creative Commons Attribution 4.0 International License (CC BY 4.0). This allows others to share the work with an acknowledgment of the work's authorship and initial publication in this journal.This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.




