Genetic diversity of Blastocystis spp: update on virulence and pathogenicity
DOI:
https://doi.org/10.70099/BJ/2024.01.04.10Keywords:
Blastocystis spp, diagnostic, pathogenicity, subtypes, virulenceAbstract
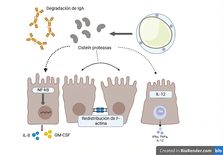
lastocystis spp. is an intestinal parasite with broad genetic diversity, highlighting four subtypes related to gastrointestinal and extraintestinal symptomatology: ST1, ST2, ST3, and ST4. Multiple studies describe the mechanisms of virulence, pathogenicity, and resistance that debate its controversial commensalism: immune evasion by sIgA degradation, cell damage mediated by cysteine proteases, and resistance to chemical/pharmacological agents. The article compiles updated information on the abovementioned mechanisms, genotypes, and diagnostic methods. Understanding the biological behavior of this protozoan is necessary to improve the diagnostic and therapeutic approach: molecular characterization and implementation of cellular assays that evaluate the activity of Blastocystis spp. Subtypes in the human intestinal tract may contribute to understanding their pathogenic activity.
References
1. del Coco VF, Molina NB, Basualdo JA, Córdoba MA. Blastocystis spp.: Advances, controversies and future challenges. Rev Argent Microbiol. 2017;49(1):110–8.
2. Pérez MR, Yáñez CM, Hernández AM, Sustaita JJD, Jiménez EG, Andrade MR, et al. Blastocystis infection frequency and subtype distribution in university students. Heliyon. 2020;6(12):0–5.
3. Paulos S, Köster PC, de Lucio A, Hernández-de-Mingo M, Cardona GA, Fernández-Crespo JC, et al. Occurrence and subtype distribution of Blastocystis sp. in humans, dogs and cats sharing household in northern Spain and assessment of zoonotic transmission risk. Zoonoses Public Health. 2018;65(8):993–1002.
4. Tapia-Veloz E, Gozalbo M, Guillén M, Dashti A, Bailo B, Köster PC, et al. Prevalence and associated risk factors of intestinal parasites among schoolchildren in Ecuador, with emphasis on the molecular diversity of Giardia duodenalis, Blastocystis sp. and Enterocytozoon bieneusi. PLoS Negl Trop Dis. 2023;17(5):1–21.
5. Sardinha-Silva A, Alves-Ferreira EVC, Grigg ME. Intestinal immune responses to commensal and pathogenic protozoa. Front Immunol. 2022;13(September):1–17.
6. Argüello-García R, Carrero JC, Ortega-Pierres MG. Extracellular Cysteine Proteases of Key Intestinal Protozoan Pathogens—Factors Linked to Virulence and Pathogenicity. Int J Mol Sci. 2023;24(16).
7. Aykur M, Malatyalı E, Demirel F, Cömert-Koçak B, Gentekaki E, Tsaousis AD, et al. Blastocystis: A Mysterious Member of the Gut Microbiome. Microorganisms. 2024;12(3):1–19.
8. Deng L, Wojciech L, Gascoigne NRJ, Peng G, Tan KSW. New insights into the interactions between Blastocystis, the gut microbiota, and host immunity. PLoS Pathog [Internet]. 2021;17(2):1–15. Available from: http://dx.doi.org/10.1371/journal.ppat.1009253
9. Kim MJ, Lee YJ, Kim TJ, Won EJ. Gut microbiome profiles in colonizations with the enteric protozoa blastocystis in korean populations. Microorganisms. 2022;10(1):1–9.
10. Deng L, Lee JWJ, Tan KSW. Infection with pathogenic Blastocystis ST7 is associated with decreased bacterial diversity and altered gut microbiome profiles in diarrheal patients. Parasites and Vectors [Internet]. 2022;15(1):1–9. Available from: https://doi.org/10.1186/s13071-022-05435-z
11. Martín-Escolano R, Ng GC, Tan KSW, Stensvold CR, Gentekaki E, Tsaousis AD. Resistance of Blastocystis to chlorine and hydrogen peroxide. Parasitol Res. 2023;122(1):167–76.
12. Rajamanikam A, Hooi HS, Kudva M, Samudi C, Kumar S. Resistance towards metronidazole in Blastocystis sp.: A pathogenic consequence. PLoS One. 2019;14(2):1–16.
13. Karamati SA, Mirjalali H, Niyyati M, Rezaei Riabi T, Yadegar A, Asadzadeh Aghdaei H, et al. Comprehensive study of phenotypic and growth rate features of blastocystis subtypes 1-3 and 6 in symptomatic and asymptomatic subjects. Iran J Parasitol. 2019;14(2):204–13.
14. Ysea MAV, Umaña MC, Fuentes SP, Campos IV, Carmona MC. Standardization of molecular techniques for the detection and characterization of intestinal protozoa and other pathogens in humans. J Venom Anim Toxins Incl Trop Dis. 2022;28:1–14.
15. Mei X, Wei L, Su C, Yang Z, Tian X, Zhang Z, et al. Advances in the axenic isolation methods of Blastocystis sp. and their applications. Parasitology. 2024;151(2):125–34.
16. Ragavan ND, Govind SK, Chye TT, Mahadeva S. Phenotypic variation in Blastocystis sp. ST3. Parasites and Vectors. 2014;7(1):2–11.
17. Nemati S, Falahati Anbaran M, Mohammad Rahimi H, Hosseini MS, Aghaei S, Khalili N, et al. Evolutionary and phylogenetic analyses of the barcoding region suggest geographical relationships among Blastocystis sp., ST3 in humans. Infect Genet Evol [Internet]. 2021;96(February):105151. Available from: https://doi.org/10.1016/j.meegid.2021.105151
18. Jiménez P, Muñoz M, Ramírez JD. An update on the distribution of Blastocystis subtypes in the Americas. Heliyon. 2022;8(12).
19. Rojas-Velázquez L, Morán P, Serrano-Vázquez A, Portillo-Bobadilla T, González E, Pérez-Juárez H, et al. The regulatory function of Blastocystis spp. on the immune inflammatory response in the gut microbiome. Front Cell Infect Microbiol. 2022;12(August):1–9.
20. Khaled S, Gantois N, Ly AT, Senghor S, Even G, Dautel E, et al. Prevalence and subtype distribution of blastocystis sp. In senegalese school children. Microorganisms. 2020;8(9):1–17.
21. Khademvatan S, Masjedizadeh R, Yousefi-Razin E, Mahbodfar H, Rahim F, Yousefi E, et al. PCR-based molecular characterization of Blastocystis hominis subtypes in southwest of Iran. J Infect Public Health [Internet]. 2018;11(1):43–7. Available from: https://doi.org/10.1016/j.jiph.2017.03.009
22. Kumarasamy V, Anbazhagan D, Subramaniyan V, Vellasamy S. Blastocystis sp., Parasite Associated with Gastrointestinal Disorders: An Overview of its Pathogenesis, Immune Modulation and Therapeutic Strategies. Curr Pharm Des. 2018;24(27):3172–5.
23. Jiménez PA, Jaimes JE, Ramírez JD. A summary of Blastocystis subtypes in North and South America. Parasites and Vectors [Internet]. 2019;12(1):1–9. Available from: https://doi.org/10.1186/s13071-019-3641-2
24. Hernández PC, Maloney JG, Molokin A, George NS, Morales L, Chaparro-Olaya J, et al. Exploring Blastocystis genetic diversity in rural schoolchildren from Colombia using next-generation amplicon sequencing reveals significant associations between contact with animals and infection risk. Parasitol Res. 2023;122(7):1451–62.
25. Ramírez JD, Sánchez A, Hernández C, Flórez C, Bernal MC, Giraldo JC, et al. Geographic distribution of human Blastocystis subtypes in South America. Infect Genet Evol [Internet]. 2016;41:32–5. Available from: http://dx.doi.org/10.1016/j.meegid.2016.03.017
26. Skotarczak B. Genetic diversity and pathogenicity of blastocystis. Ann Agric Environ Med. 2018;25(3):411–6.
27. Das R, Khalil S, Mirdha BR, Makharia GK, Dattagupta S, Chaudhry R. Molecular characterization and subtyping of blastocystis species in irritable bowel syndrome patients from north India. PLoS One. 2016;11(1):1–9.
28. Fontanelli Sulekova L, Gabrielli S, Furzi F, Milardi GL, Biliotti E, De Angelis M, et al. Molecular characterization of Blastocystis subtypes in HIV-positive patients and evaluation of risk factors for colonization. BMC Infect Dis. 2019;19(1):1–7.
29. Ajjampur SSR, Png CW, Chia WN, Zhang Y, Tan KSW. Ex vivo and in vivo mice models to study blastocystis spp. adhesion, colonization and pathology: Closer to proving koch’s postulates. PLoS One. 2016;11(8):1–17.
30. Salvador F, Sulleiro E, Sánchez-Montalvá A, Alonso C, Santos J, Fuentes I, et al. Epidemiological and clinical profile of adult patients with Blastocystis sp. infection in Barcelona, Spain. Parasites and Vectors [Internet]. 2016;9(1):1–7. Available from: http://dx.doi.org/10.1186/s13071-016-1827-4
31. El Safadi D, Cian A, Nourrisson C, Pereira B, Morelle C, Bastien P, et al. Prevalence, risk factors for infection and subtype distribution of the intestinal parasite Blastocystis sp. from a large-scale multi-center study in France. BMC Infect Dis [Internet]. 2016;16(1):1–11. Available from: http://dx.doi.org/10.1186/s12879-016-1776-8
32. Sheela DS, Chandramathi S, Suresh K. Epitope variances demonstrated by blastocystis sp. ST3 symptomatic and asymptomatic isolates. Trop Biomed. 2020;37(1):210–7.
33. Bahrami F, Babaei E, Badirzadeh A, Riabi TR, Abdoli A. Blastocystis, urticaria, and skin disorders: review of the current evidences. Eur J Clin Microbiol Infect Dis. 2020;39(6):1027–42.
34. Rajamanikam A, Isa MNM, Samudi C, Devaraj S, Govind SK. Gut bacteria influence Blastocystis sp. phenotypes and may trigger pathogenicity. PLoS Negl Trop Dis [Internet]. 2023;17(3):1–20. Available from: http://dx.doi.org/10.1371/journal.pntd.0011170
35. Mayneris-Perxachs J, Arnoriaga-Rodríguez M, Garre-Olmo J, Puig J, Ramos R, Trelis M, et al. Presence of Blastocystis in gut microbiota is associated with cognitive traits and decreased executive function. ISME J. 2022;16(9):2181–97.
36. Huang LS, Yeh YM, Chiu SF, Huang PJ, Chu LJ, Huang CY, et al. Intestinal microbiota analysis of different Blastocystis subtypes and Blastocystis-negative individuals in Taiwan. Biomed J [Internet]. 2023;(October):100661. Available from: https://doi.org/10.1016/j.bj.2023.100661
37. Castañeda S, Muñoz M, Villamizar X, Hernández PC, Vásquez LR, Tito RY, et al. Microbiota characterization in Blastocystis-colonized and Blastocystis-free school-age children from Colombia. Parasites and Vectors [Internet]. 2020;13(1):1–12. Available from: https://doi.org/10.1186/s13071-020-04392-9
38. Marangi M, Boughattas S, De Nittis R, Pisanelli D, delli Carri V, Lipsi MR, et al. Prevalence and genetic diversity of Blastocystis sp. among autochthonous and immigrant patients in Italy. Microb Pathog [Internet]. 2023;185(October):106377. Available from: https://doi.org/10.1016/j.micpath.2023.106377
39. Lhotská Z, Jirků M, Hložková O, Brožová K, Jirsová D, Stensvold CR, et al. A Study on the Prevalence and Subtype Diversity of the Intestinal Protist Blastocystis sp. in a Gut-Healthy Human Population in the Czech Republic. Front Cell Infect Microbiol. 2020;10(October):1–14.
40. Rudzińska M, Kowalewska B, Wąż P, Sikorska K, Szostakowska B. Blastocystis subtypes isolated from travelers and non-travelers from the north of Poland – A single center study. Infect Genet Evol [Internet]. 2019;75(June):103926. Available from: https://doi.org/10.1016/j.meegid.2019.103926
41. Ben Abda I, Maatoug N, Ben Romdhane R, Bouhelmi N, Zallegua N, Aoun K, et al. Prevalence and subtype identification of Blastocystis sp. in healthy individuals in the Tunis Area, Tunisia. Am J Trop Med Hyg. 2017;96(1):202–4.
42. Naguib D, Gantois N, Desramaut J, Arafat N, Mandour M, Abdelmaogood AKK, et al. Molecular Epidemiology and Genetic Diversity of the Enteric Protozoan Parasite Blastocystis sp. in the Northern Egypt Population. Pathogens. 2023;12(11):1–13.
43. Guilavogui T, Gantois N, Even G, Desramaut J, Dautel E, Denoyelle C, et al. Detection, Molecular Identification and Transmission of the Intestinal Protozoa Blastocystis sp. in Guinea from a Large‐Scale Epidemiological Study Conducted in the Conakry Area. Microorganisms. 2022;10(2).
44. Nemati S, Zali MR, Johnson P, Mirjalali H, Karanis P. Molecular prevalence and subtype distribution of Blastocystis sp. In Asia and in Australia. J Water Health. 2021;19(5):687–704.
45. Kapczuk P, Kosik-Bogacka D, Kupnicka P, Metryka E, Simińska D, Rogulska K, et al. The influence of selected gastrointestinal parasites on apoptosis in intestinal epithelial cells. Biomolecules. 2020;10(5).
46. Nourrisson C, Wawrzyniak I, Cian A, Livrelli V, Viscogliosi E, Delbac F, et al. On Blastocystis secreted cysteine proteases: A legumain-activated cathepsin B increases paracellular permeability of intestinal Caco-2 cell monolayers. Parasitology. 2016;143(13):1713–22.
47. Gonzalez-Arenas NR, Villalobos G, Vargas-Sanchez GB, Avalos-Galarza CA, Marquez-Valdelamar LM, Ramirez-Miranda ME, et al. Is the genetic variability of Cathepsin B important in the pathogenesis of Blastocystis spp.? Parasitol Res. 2018;117(12):3935–43.
48. Lee GO, Whitney HJ, Blum AG, Lybik N, Cevallos W, Trueba G, et al. Household coping strategies associated with unreliable water supplies and diarrhea in Ecuador, an upper-middle-income country. Water Res [Internet]. 2020;170:115269. Available from: https://doi.org/10.1016/j.watres.2019.115269
49. Franklin F, Rajamanikam A, Raju CS, Gill JS, Francis B, Sy-Cherng LW, et al. Higher amoebic and metronidazole resistant forms of Blastocystis sp. seen in schizophrenic patients. Parasites and Vectors [Internet]. 2022;15(1):1–16. Available from: https://doi.org/10.1186/s13071-022-05418-0
50. Nourrisson C, Brunet J, Flori P, Moniot M, Bonnin V, Delbac F, et al. Comparison of DNA extraction methods and real-time PCR assays for the detection of blastocystis sp. in stool specimens. Microorganisms. 2020;8(11):1–8.
Published
How to Cite
License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors who publish with BioNatura Journal agree to the following terms: Authors retain copyright and grant the BioNatura Institutional Publishing Consortium (BIPC) right of first publication with the work simultaneously licensed under a Creative Commons Attribution 4.0 International License (CC BY 4.0). This allows others to share the work with an acknowledgment of the work's authorship and initial publication in this journal.This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.




