Integrated Cerebral Autoregulation Score for Early Risk Stratification in Severe TBI

Marlon Carbonell González  1*, Rosali Santiago Roibal
1*, Rosali Santiago Roibal 

 3
3
1 High Quality Medical Services, Houston, United States, deborahcabrera0211@gmail.com
2All Behavior Community Inc., Florida, United States rosalisantiago97@gmail.com
3 Emergency Medicine Department, Torrevieja University Hospital, Alicante Province, Valencian Community, Spain,
ayalaperezjorgeluis@gmail.com
Corresponding author: marloncarbonell95@gmail.com
ABSTRACT
In
a scenario where precise tools are crucial, a 10-year-old boy arrives at the
emergency room with a Glasgow Coma Scale of 5 following a car accident,
requiring immediate vasopressor support. This situation highlights the urgent
need for effective tools in pediatric neurocritical care. Impaired cerebral
autoregulation is associated with poor outcomes in severe pediatric traumatic
brain injury (TBI), yet integrated bedside assessment tools are lacking. We
conducted a retrospective multicenter cohort study including 100 children (ages
0–18) with severe TBI (Glasgow Coma Scale ≤8) who required vasopressor support
within 72 hours of admission. Our objective was to identify early predictors of
autoregulatory failure and develop the Pediatric Integrated Cerebral Autoregulation
Score (pICAS). Multimodal neuromonitoring included intracranial pressure (ICP),
cerebral perfusion pressure (CPP), pressure reactivity index (PRx), and mean
arterial pressure (MAP) variability. The primary outcome was poor clinical
result (mortality or PGOS ≤3), observed in 46% of patients. The pICAS was
created to predict clinical deterioration by integrating PRx and other
physiological variables. Multivariate logistic regression identified five early
predictors for the pICAS (score range 0–5): ICP ≥20 mmHg, PRx >0.30 for
>35% of monitoring time, MAP variability >12 mmHg, CPP below optimal for
>30% of the time, and norepinephrine-equivalent dose ≥0.1 µg/kg/min. The
pICAS demonstrated higher accuracy (AUC 0.89, 95% CI 0.83–0.95) than ICP alone
(AUC 0.72) or PRx alone (AUC 0.76) (p < 0.01), and showed excellent
calibration (Hosmer-Lemeshow p=0.62) with internal validation
(optimism-corrected AUC 0.87). Risk of impaired autoregulation increased from
10% (score 0–1) to 90% (score 5). At a cutoff of ≥3, sensitivity was 85%,
specificity was 82%, PPV was 77%, and NPV was 88%. The pICAS offers a practical
framework for early risk stratification and individualized management.
Keywords: pediatric
traumatic brain injury; cerebral autoregulation; pressure reactivity index;
pediatric integrated cerebral autoregulation score (pICAS); risk
stratification; prediction model; pediatric neurocritical care.
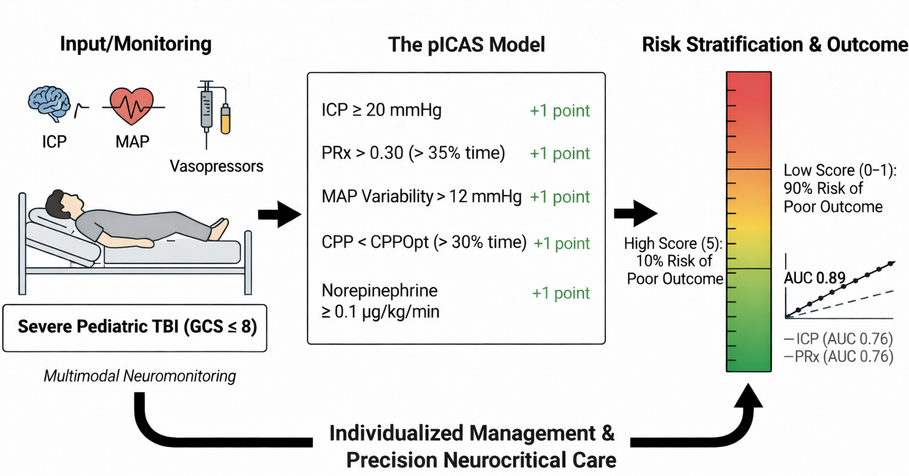
Graphical
Abstract. Schematic
representation of the Pediatric Integrated Cerebral Autoregulation Score
(pICAS) for early risk stratification in severe pediatric traumatic brain
injury. Multimodal neuromonitoring variables—including intracranial
pressure, pressure reactivity index, mean arterial pressure variability,
cerebral perfusion pressure relative to optimal targets, and vasopressor
requirement—are integrated into a five-point score. Increasing scores
indicate a higher probability of impaired cerebral autoregulation and poor
clinical outcome, supporting individualized management and precision
neurocritical care.
INTRODUCTION
Severe
traumatic brain injury (TBI) is a leading cause of death and long-term
neurological disability in children worldwide. It accounts for a large share of
pediatric neurocritical care admissions and health resource use 1,2,3. While the primary mechanical injury is mostly irreversible,
secondary brain injury—due to intracranial hypertension, cerebral
hypoperfusion, metabolic disturbances, and disordered cerebrovascular
control—is a critical, potentially modifiable determinant of outcome 4,5,6.
Cerebral
autoregulation is a key mechanism that maintains stable cerebral blood flow
across a wide range of blood pressures. In pediatric severe TBI, autoregulation
is often impaired. As a result, cerebral perfusion pressure (CPP) depends
heavily on systemic hemodynamics and vasopressor therapy 7,8,9. Loss of autoregulation leaves the developing brain
vulnerable: ischemia can occur during hypotension, and hyperemia and edema can
develop during hypertension.
The
pressure reactivity index (PRx), derived from the correlation between slow
waves in mean arterial pressure (MAP) and intracranial pressure (ICP), has
emerged as a validated, continuous, bedside marker of cerebrovascular
reactivity and outcome in pediatric TBI 10,11,12. PRx-based approaches allow estimation of individualized
optimal CPP (CPPopt), highlighting the heterogeneity of perfusion requirements
across children and over time. Importantly, impaired autoregulation rarely
occurs in isolation; it reflects the interaction of systemic hemodynamic
instability, vasopressor exposure, intracranial compliance, cerebral
oxygenation, and metabolic stress unique to pediatric physiology 13.
Rationale and Knowledge Gap
Despite
the growing use of multimodal neuromonitoring, including ICP, CPP, PRx, and
brain tissue oxygen tension (PbtO₂), current pediatric TBI management relies
mainly on population-based MAP and CPP targets. These targets do not consider
individual autoregulatory capacity or its interaction with systemic
hemodynamics 14,15. Clinicians today face significant uncertainty in achieving
optimal CPP targets, as these are typically derived from generalized data
rather than personalized patient metrics. The Pediatric Integrated Cerebral
Autoregulation Score (pICAS) offers an opportunity to redefine this standard by
enabling individualized assessment and management of cerebral perfusion in
children. While vasopressor therapy is essential to maintain CPP, it may worsen
cerebral perfusion instability in children with impaired autoregulation by
increasing MAP variability and encouraging pressure-passive cerebral blood flow
16.
No
integrated bedside tool combines available cerebral and systemic data to help
quickly identify children at high risk for autoregulatory failure and poor
outcomes. Previous studies focused on single variables or domains, limiting
personalized bedside use 17,18,19,20.
We
hypothesized that impaired cerebral autoregulation in pediatric severe TBI
reflects a multidimensional interplay among intracranial dynamics,
individualized CPP targets, and systemic hemodynamic instability. We conducted
a retrospective observational study of 100 children with severe TBI requiring
vasopressor support and continuous multimodal neuromonitoring to develop and
internally validate the Pediatric Integrated Cerebral Autoregulation Score
(pICAS), a bedside tool designed to stratify risk of impaired autoregulation,
neurological deterioration, and PICU mortality.
METHODS
Study Design and Setting
This
retrospective, multicenter observational cohort study included 100 consecutive
children with severe traumatic brain injury (TBI) admitted to tertiary-level
Pediatric Neurocritical Care Units between January 2018 and December 2023 (Fig.
1). The centers participating in this study represented a diverse range of
geographic regions, including urban centers in Europe and Latin America, which
provided access to varied demographic populations. The study sites ranged in
resource availability, with some having comprehensive multimodal monitoring
capabilities while others operated with more limited resources. The study was
conducted and reported in accordance with the TRIPOD guidelines for prediction
model development and the STROBE checklist for observational studies.
Institutional Review Board approval was obtained at all participating centers,
with a waiver of informed consent due to the retrospective design and use of
anonymized data, in compliance with the Declaration of Helsinki. By adhering to
these guidelines, we aim to reassure reviewers about the rigor and transparency
of our methodological approach.
Physiological
signals were measured at high speed (100 times per second) and analyzed using
ICM+ software (a system for processing brain monitoring data). The pressure
reactivity index (PRx) was calculated as a moving Pearson correlation
coefficient (a measure of how closely two variables are related) between mean
arterial pressure (MAP, average blood pressure) and intracranial pressure (ICP,
pressure inside the skull). This was done every minute, based on 30 samples,
each averaging 10 seconds, with the data set updated every minute and using a
50% overlapping window for more accuracy. Individual optimal cerebral perfusion
pressure (CPPopt) was determined using a multi-window, weighted parabolic fit
algorithm (a mathematical method to find the best CPP for each child) applied
over a rolling 4-hour period, provided at least half of the data points were
present to ensure accuracy.
Flow diagram
illustrating the selection process for the pediatric severe traumatic brain
injury (TBI) cohort. Of 125 screened patients, 100 met eligibility criteria and
were included in the final analysis, stratified by cerebral autoregulation
status.
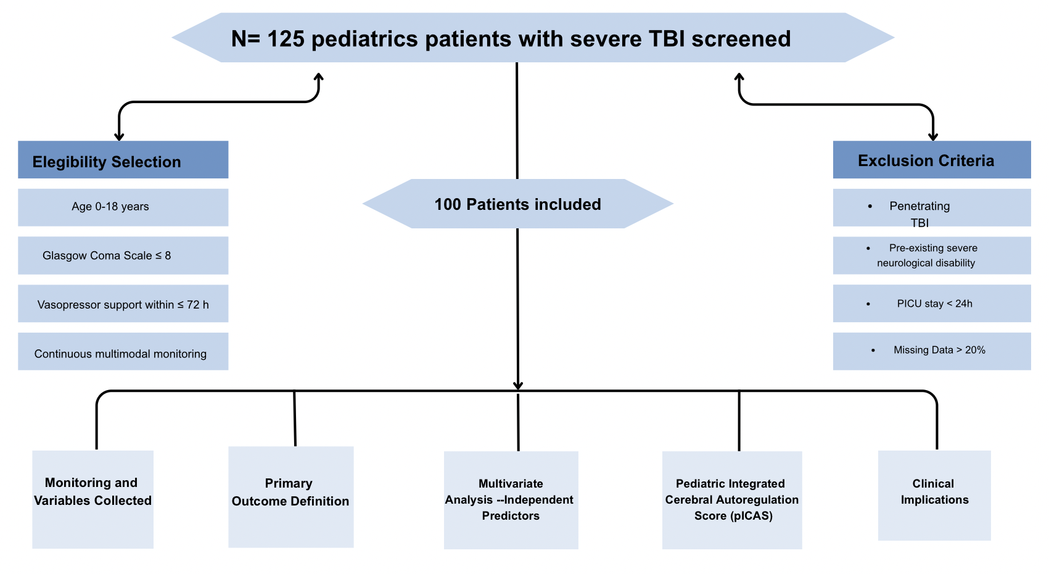
Abbreviations: PICU:
Pediatric Intensive Care Unit; pICAS: Pediatric Integrated Cerebral
Autoregulation Score; TBI: Traumatic Brain Injury.
Figure
1. Flow diagram of the patient selection process for the pediatric severe
traumatic brain injury (TBI) cohort.
Patient Selection
Children
aged 0–18 years with severe TBI (Glasgow Coma Scale ≤8 after initial
resuscitation) were eligible if they required vasopressor support within the
first 72 hours of Pediatric Intensive Care Unit (PICU) admission and underwent
continuous ICP and cerebral autoregulation monitoring.
Patients
were excluded based on the following criteria:
- Penetrating TBI
- Pre-existing severe neurological disability (baseline Pediatric Glasgow Outcome Scale ≤3)
- PICU length of stay: 24 hours
- Absence of continuous multimodal neuromonitoring
- Missing data >20% for key physiological variables
Multimodal Cerebral Monitoring
ICP
was continuously monitored using special devices: either probes placed in the
brain tissue or cerebrospinal fluid drainage (external ventricular drainage),
both set up according to hospital protocols for children. MAP (mean arterial
pressure, or average blood pressure) was measured invasively using an arterial
catheter. CPP (cerebral perfusion pressure) was calculated as the difference
between MAP and ICP, using age-based normal values 21,22.
Cerebral
autoregulation was assessed using the pressure reactivity index (PRx), a
measure of the temporal relationship between changes in mean arterial pressure
(MAP) and intracranial pressure (ICP). Impaired autoregulation was defined as an
average PRx greater than 0.30 during the patient's monitoring period 23.
Individualized
optimal CPP (CPPopt) was estimated using established PRx-based algorithms, and
CPP deviation from CPPopt was quantified as the proportion of monitored time
below the individualized optimal range. Brain tissue oxygen tension (PbtO₂) was
recorded when available, with cerebral hypoxia defined as PbtO₂ < 20 mmHg
for >10% of monitoring duration, a threshold commonly extrapolated from
adult neurocritical care and previously applied in pediatric TBI studies 24.
Systemic Hemodynamic and Metabolic Variables
Systemic
hemodynamics included mean, minimum, and variability (standard deviation) of
MAP, heart rate, and central venous pressure when available. Vasopressor
therapy was characterized by agent type, maximum and cumulative dose (expressed
as norepinephrine-equivalent µg/kg/min), and duration 25,26,27.
Metabolic
stress was assessed via serum lactate levels at PICU admission and 24 hours,
and lactate clearance over the first 24 hours. Episodes of hypotension and MAP
variability were quantified to evaluate hemodynamic instability 28,29,30.
Outcome Measures
The
primary outcome was redefined as a poor clinical outcome, a composite endpoint
comprising PICU mortality or an unfavorable neurological status at hospital
discharge (Pediatric Glasgow Outcome Scale ≤ 3). This change was implemented to
evaluate the pICAS as a prognostic tool and to avoid mathematical circularity,
as PRx is now treated strictly as a predictor of clinical deterioration rather
than the primary endpoint itself.
Development of the Pediatric Integrated Cerebral
Autoregulation Score (pICAS)
Candidate
variables were selected a priori based on physiological plausibility and prior
literature, encompassing cerebral variables such as ICP burden, PRx, CPPopt
deviation, and PbtO₂ hypoxia burden; hemodynamic variables including MAP
variability, vasopressor dose, and duration; and metabolic variables such as
lactate levels and clearance. To eliminate mathematical circularity and ensure
clinical relevance, the final pICAS was constructed using five independent
predictors of poor clinical outcome, defined as PICU mortality or unfavorable
neurological status at discharge, identified through multivariate logistic
regression. Notably, lactate levels were retained in the final score over PbtO₂
due to their stronger association with systemic perfusion deficits and clinical
outcomes. These selected predictors are clinically actionable, as they can
guide targeted interventions within management protocols. These components
include an ICP burden ≥ 20 mmHg, a PRx > 0.30 for > 35% of monitoring
time, a CPP deviation from CPPopt > 30% of monitoring time, a MAP
variability > 12 mmHg, and a norepinephrine-equivalent dose ≥ 0.1 µg/kg/min.
Each predictor was assigned 1 point, yielding a total pICAS score ranging from
0 to 5, with higher scores indicating a greater risk of adverse clinical
outcomes.
Statistical Analysis
Continuous
variables are presented as median (interquartile range, IQR), and categorical
variables as counts (percentages). Univariate comparisons used the Mann–Whitney
U test or chi-square test, as appropriate. Variables with p < 0.10 in
univariate analysis were entered into a multivariate logistic regression with
backward stepwise elimination to identify independent predictors of impaired
autoregulation. In addition to excluding patients with more than 20% missing
data, smaller gaps in secondary physiological variables were addressed using
Multiple Imputation by Chained Equations (MICE). For these imputations,
variables such as serum lactate levels, MAP variability, and norepinephrine
doses were included. A total of 10 imputations were conducted, and diagnostics
verified the accuracy and reliability of the imputed datasets. Overall, 6% of
all candidate values were imputed, helping readers assess the potential impact
on the analysis. To ensure the robustness of the pICAS, a sensitivity analysis
was conducted comparing the complete-case cohort with the imputed dataset,
which showed no significant differences in the model's coefficients or AUC
(0.89 vs 0.88, p=0.45).
Cutoff
values for continuous predictors included in the Pediatric Integrated Cerebral
Autoregulation Score (pICAS) were determined a priori based on physiological
relevance and further refined using receiver operating characteristic (ROC)
analysis, selecting thresholds that maximized the Youden index for predicting
impaired cerebral autoregulation. The MAP variability threshold represents a
high-risk extreme of hemodynamic instability rather than the population median,
consistent with its intended use in identifying clinically relevant
autoregulatory failure.
Model
performance was assessed using the area under the receiver operating
characteristic curve (AUC) for discrimination, the Hosmer–Lemeshow test for
calibration, and the Nagelkerke pseudo-R² for overall performance. Internal
validation employed 1,000 bootstrap resamples to evaluate model stability and
optimism-corrected performance. Statistical significance was set at p = 0.05
(two-tailed). Notably, while the AUC provides a measure of statistical
discrimination, the clinical utility becomes apparent when considering the
real-world impact of a pICAS cutoff of 3. Such a threshold can fundamentally
alter therapy by prioritizing more intensive hemodynamic monitoring and
customized interventions for those patients identified at heightened risk of
clinical deterioration 31,32.
RESULTS
Study Population
A
total of 100 pediatric patients with severe traumatic brain injury (TBI)
requiring vasopressor support were included in the analysis (median age 9
years, IQR 4–14; 63% male). The predominant mechanisms of injury were falls
(45%), motor vehicle collisions (38%), and non-accidental trauma/assaults
(17%). At PICU admission, the median Pediatric Glasgow Coma Scale (GCS) score
was 6 (IQR 5–7).
Overall,
49 patients (49%) developed impaired cerebral autoregulation (mean PRx
>0.30), 24 patients (24%) died during PICU stay, and 46 patients (46%) had
an unfavorable neurological outcome at hospital discharge (Pediatric Glasgow
Outcome Scale ≤3).
Baseline
demographic, injury, and hemodynamic characteristics of the cohort are
summarized in Table 1. Patients with impaired autoregulation had higher ICP,
MAP variability, norepinephrine requirements, and lactate levels, and
experienced worse neurological outcomes compared with those with preserved
autoregulation.
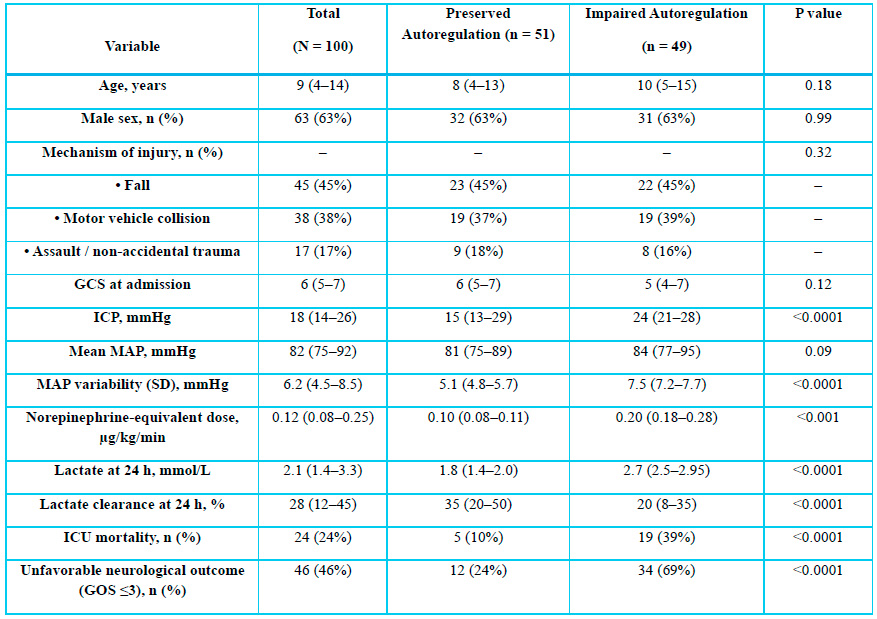
Baseline
characteristics by cerebral autoregulation status (N=100). Data are median
(IQR) or n (%). Comparisons by Mann–Whitney U or chi-square test; p 0.05
considered significant.
Abbreviations: GCS,
Glasgow Coma Scale; ICP, intracranial pressure; MAP, mean arterial pressure;
ICU, intensive care unit; GOS, Glasgow Outcome Scale.
Table
1. Baseline Demographic, Injury, and Hemodynamic Characteristics According to
Cerebral Autoregulation Status.
Cerebral and Hemodynamic Characteristics
Children
with impaired autoregulation exhibited significant differences in both cerebral
and systemic hemodynamic parameters compared with those with preserved
autoregulation. Specifically, the median intracranial pressure (ICP) was 24
mmHg (IQR 21–28) versus 15 mmHg (IQR 13–29) (p < 0.0001) (Fig. 2a), while
the PRx burden (PRx > 0.30) reached a median of 35% of monitoring time (IQR
34–42%) compared to 12% (IQR 10–15%) in the preserved group (p < 0.0001) (Fig.
2b). Systemic variability was also more pronounced in the impaired group, with
a MAP variability SD of 7.5 mmHg (IQR 7.2–7.7) versus 5.1 mmHg (IQR 4.8–5.7) (p
< 0.0001) (Fig. 2c). Furthermore, the duration of CPP below CPPopt was
significantly higher in affected children, occupying 38% of monitoring time
(IQR 35–43%) compared to 12% (IQR 10–15%) (p < 0.0001) (Fig. 2d).
Vasopressor
requirements were significantly higher in patients with impaired autoregulation
(median norepinephrine-equivalent dose 0.20 µg/kg/min, IQR 0.18–0.28) compared
with patients with preserved autoregulation (median 0.10 µg/kg/min, IQR 0.08–0.11;
p < 0.0001) (Fig. 2e). Brain tissue hypoxia (PbtO₂ < 20 mmHg for > 10%
of monitoring time) occurred in 21/49 patients (44%) with impaired
autoregulation versus 6/51 (12%) in the preserved group (p < 0.0001) (Fig.
2f). Serum lactate at 24 hours was also higher in the impaired autoregulation
group (median 2.7 mmol/L, IQR 2.5–2.95) compared with the preserved group
(median 1.8 mmol/L, IQR 1.4–2.0; p < 0.0001) (Fig. 2g).
Patients with
impaired cerebral autoregulation demonstrated worse physiological and metabolic
parameters compared with those with preserved autoregulation. (a) ICP: Impaired
patients showed significantly higher pressure (24 vs 15 mmHg). (b) PRx Burden:
Impaired group spent more time in pressure-passive state (35% vs 12% of time).
(c) MAP Variability: The impaired group showed greater hemodynamic instability
(SD 7.5 vs 5.1 mmHg). (d) CPP < CPPopt: Impaired patients spent more time
below optimal perfusion (38% vs 12% of time). Vasopressors: Higher doses needed
for impaired autoregulation (0.20 vs 0.10 µg/kg/min). (f) Hypoxia: Higher
incidence of brain tissue hypoxia in the impaired group (44% vs 12%). (g)
Lactate: Elevated serum lactate in impaired patients (2.7 vs 1.8 mmol/L).
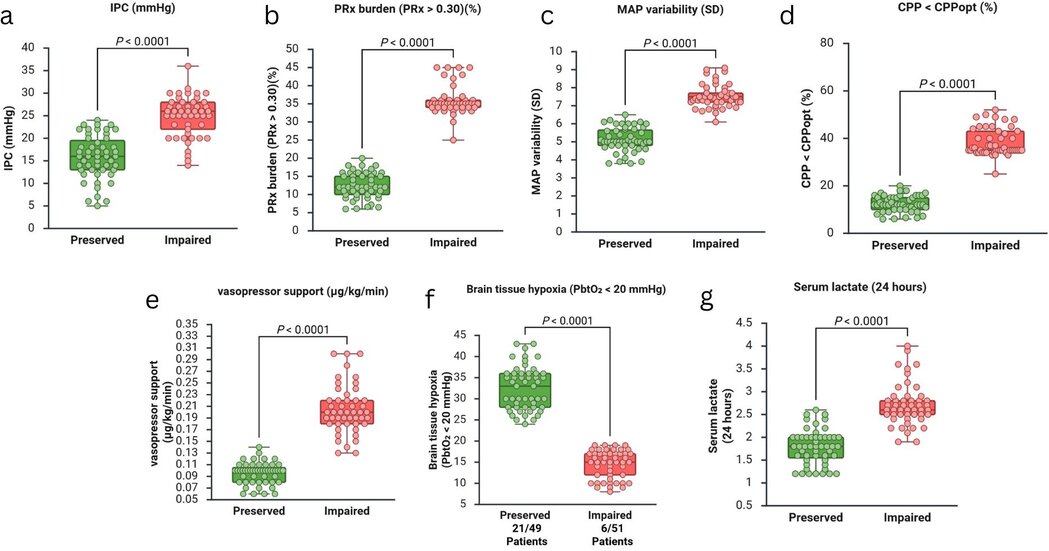
Abbreviations: ICP:
Intracranial Pressure; MAP: Mean Arterial Pressure; CPP: Cerebral Perfusion
Pressure; CPPopt: Optimal Cerebral Perfusion Pressure; SD: Standard Deviation;
PRx: Pressure Reactivity Index; PbtO₂: Brain tissue oxygen tension; IQR:
Interquartile Range.
Figure
2. Physiological and metabolic differences between patients with preserved and
impaired cerebral autoregulation in severe pediatric traumatic brain injury.
Pediatric Integrated Cerebral Autoregulation Score (pICAS)
Derivation
Multivariate
logistic regression identified five independent predictors of impaired cerebral
autoregulation, which were used to construct the Pediatric Integrated Cerebral
Autoregulation Score (pICAS) (Table 2). Each predictor was assigned 1 point,
yielding a total score of 0-5. The scoring components included ICP ≥ 20 mmHg,
PRx > 0.30 for > 35% of monitoring time, MAP variability > 12 mmHg,
CPP below CPPopt for> 30% of monitoring time, and a
norepinephrine-equivalent dose of 0.1 µg/kg/min.
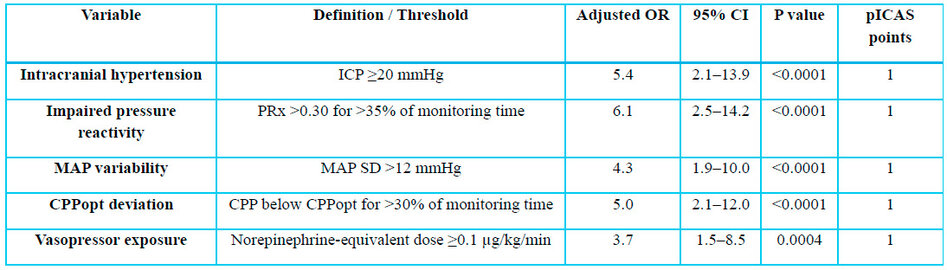
Multivariate
logistic regression identified independent predictors of impaired cerebral
autoregulation in children with severe TBI (N=100). Each predictor was assigned
1 point for the pICAS (0–5). P < 0.05 was considered significant.
Abbreviations:
OR, odds ratio; CI, confidence interval; ICP, intracranial pressure; PRx,
pressure reactivity index; MAP, mean arterial pressure; CPPopt, optimal
cerebral perfusion pressure; pICAS, Pediatric Integrated Cerebral
Autoregulation Score.
Table 2. Multivariate Logistic Regression Model for
Impaired Cerebral Autoregulation and pICAS Derivation.
Model Performance and Risk Stratification
To demonstrate the incremental value of the pICAS, we
compared its discriminative ability with that of isolated monitoring variables.
The pICAS showed significantly higher accuracy (AUC 0.89, 95% CI 0.83–0.95) for
predicting poor clinical outcome, defined as PICU mortality or unfavorable
neurological status at discharge, compared to ICP alone (AUC 0.72) or PRx alone
(AUC 0.76) (p < 0.01). This confirms that integrating systemic hemodynamics
with individualized autoregulation metrics provides superior risk stratification
compared with traditional population-based targets. The distribution of the
cohort according to the pICAS was: score 0–1 (n=22), score 2 (n=18), score 3
(n=20), score 4 (n=25), and score 5 (n=15). In addition to the Hosmer–Lemeshow
test (p=0.62), a calibration plot showed strong alignment between the predicted
probability of poor clinical outcome and the observed event rate across all
risk deciles, with a slight overestimation in the highest risk category (score
5).
Furthermore, the observed incidence of poor clinical outcome
increased in a stepwise manner with increasing pICAS score. Patients with a
score of 0-1 showed an incidence of 10%, which increased to 25% for a score of
2, 55% for a score of 3, 78% for a score of 4, and reached 90% for a score of 5
(Fig. 3). At a cutoff of ≥ 3 points, the pICAS demonstrated sensitivity of 85%,
specificity of 82%, positive predictive value of 77%, and negative predictive
value of 88%. The model showed excellent discrimination and adequate
calibration, with a Hosmer–Lemeshow p-value of 0.62 (Fig. 4). Multimodal
neuromonitoring parameters and the associated risk of poor clinical outcome
across pICAS categories are presented in Table 3.
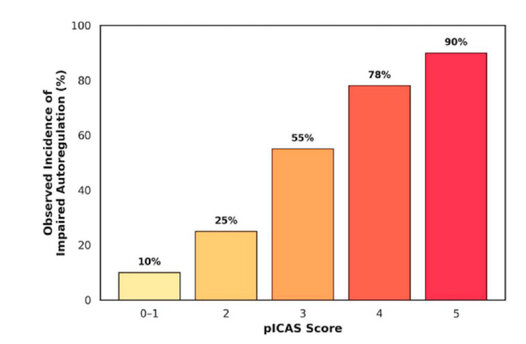
The bar chart shows a stepwise
increase in the observed incidence of impaired autoregulation (mean PRx >
0.30) as the pICAS score increases from 0 to 5.
Abbreviations: pICAS: Pediatric Integrated Cerebral Autoregulation Score;
PRx: Pressure Reactivity Index. Data labels indicate the percentage of patients
within each score tier.
Figure 3. Risk
stratification according to the Pediatric Integrated Cerebral Autoregulation
Score
(pICAS).
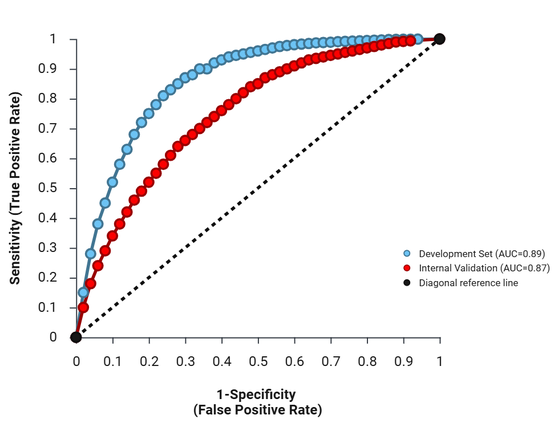
The blue curve represents the
Development Set (AUC = 0.89), while the red curve shows the Internal Validation
(AUC = 0.87). Both curves demonstrate strong predictive accuracy, remaining
well above the diagonal reference line representing chance-level performance
(AUC = 0.50).
Abbreviations: AUC: Area Under the Curve; ROC: Receiver Operating
Characteristic; Sensitivity: True Positive Rate (TPR); 1-Specificity: False
Positive Rate (FPR)
Figure 4. Receiver
Operating Characteristic (ROC) curves evaluate model performance.
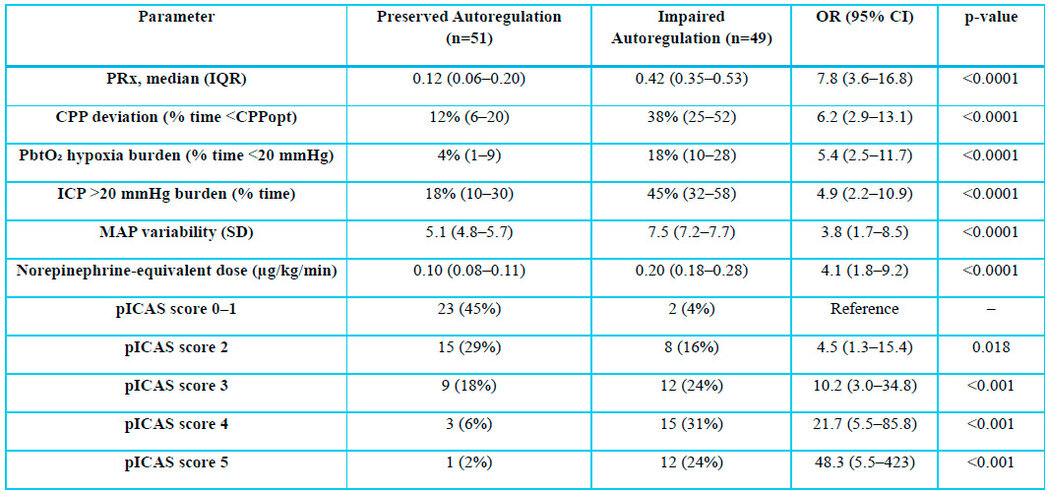
Comparison of neuromonitoring
parameters and pICAS categories between patients with preserved and impaired
cerebral autoregulation. Odds ratios represent the risk of impaired autoregulation;
pICAS 0–1 used as a reference. P < 0.05 is considered significant.
Abbreviations: OR, odds ratio; CI, confidence interval; PRx, pressure
reactivity index; CPPopt, optimal cerebral perfusion pressure; PbtO₂, brain
tissue oxygen tension; ICP, intracranial pressure; MAP, mean arterial pressure;
pICAS, Integrated Cerebral Autoregulation Score.
Table 3. Multimodal
Physiologic Parameters and Performance of the Integrated Cerebral
Autoregulation Score (pICAS).
Internal Validation
Internal validation using 1,000 bootstrap resamples confirmed
model stability, yielding an optimism-corrected AUC of 0.87, indicating robust
performance and reproducibility. Subgroup analyses demonstrated consistent
predictive performance across age groups (infants, children, adolescents), sex,
and injury mechanisms, supporting the generalizability of the pICAS within a
pediatric severe TBI population requiring vasopressors. Furthermore, to address
the application of pICAS in resource-limited PICUs, we propose several adaptation
strategies. Firstly, simpler variables, such as easily measurable physiological
parameters, could be used in place of complex neuromonitoring data. Secondly,
adjusted thresholds that align with the available data in these settings can be
established to ensure accurate predictions. These adaptations can enable
broader implementation and enhance stakeholder engagement even with varying
levels of monitoring capacity. Future research should explore pragmatic
modifications to tailor the pICAS to diverse medical environments.
Multimodal Insights into Autoregulatory Dysfunction
Children
with impaired autoregulation in our cohort exhibited elevated ICP, prolonged
time with CPP below CPPopt, increased MAP variability, and frequent episodes of
brain tissue hypoxia (PbtO₂ 20 mmHg). These findings highlight the dynamic,
pressure-passive nature of cerebral perfusion in pediatric patients with severe
TBI. Elevated serum lactate and reduced lactate clearance indicate systemic
perfusion deficits contributing to autoregulatory failure, emphasizing the
importance of integrating both cerebral and systemic parameters for early risk
prediction. Individualized optimal CPP (CPPopt) was derived from the U-shaped
relationship between PRx and CPP (Fig. 5).
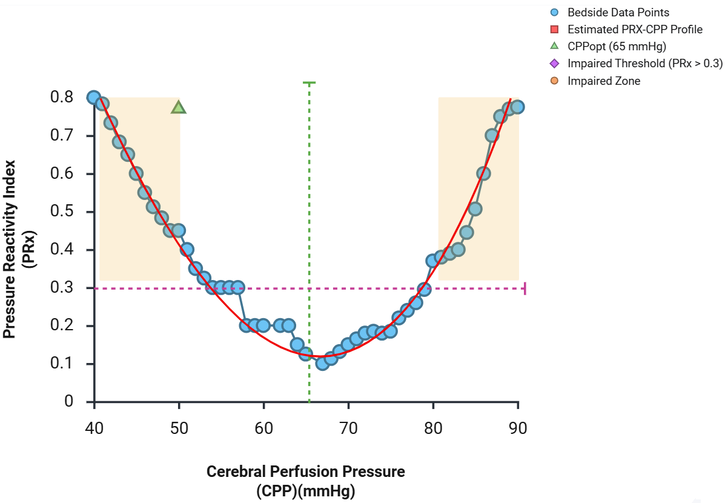
The blue circles
represent real-time bedside data points, while the red line indicates the
estimated PRx-CPP parabolic profile. The green dashed line identifies the
Optimal Cerebral Perfusion Pressure (CPPOpt) at 65 mmHg, where PRx is at its
lowest. The purple dashed line marks the Impaired Threshold (PRx > 0.3),
with shaded tan areas indicating the Impaired Zones where cerebrovascular
reactivity is compromised.
Abbreviations:
PRx: Pressure Reactivity Index; CPP: Cerebral Perfusion
Pressure; CPPopt: Optimal Cerebral Perfusion Pressure; mmHg: Millimeters of
Mercury
Figure
5. Cerebral Autoregulation Profile showing the relationship between Pressure
Reactivity Index (PRx) and Cerebral Perfusion Pressure (CPP).
DISCUSSION
Principal Findings
This
study demonstrates that impaired cerebral autoregulation in children with
severe traumatic brain injury (TBI) requiring vasopressor support results from
a multidimensional interplay among intracranial hypertension, systemic
hemodynamic instability, deviation from individualized optimal CPP (CPPopt),
and vasopressor exposure. The Pediatric Integrated Cerebral Autoregulation
Score (pICAS) consolidates these factors into a bedside-applicable tool for
early risk stratification of children at highest risk for neurological
deterioration and PICU mortality 33,34,35.
Vasopressor Therapy and Cerebrovascular Coupling
While
vasopressors are essential to maintain CPP, our data show that higher doses and
prolonged exposure were independently associated with impaired autoregulation.
By integrating cerebral, hemodynamic, and metabolic factors, the pICAS captures
this critical interaction, providing a practical framework to guide
individualized vasopressor titration and minimize episodes of pressure-passive
cerebral perfusion 36,37.
Clinical Implications
The
pICAS enables early identification of pediatric patients at high risk of
autoregulatory failure, supporting closer hemodynamic monitoring,
individualized CPP optimization based on CPPopt, and judicious vasopressor
management to reduce MAP variability. By combining multiple routinely available
multimodal parameters, pICAS facilitates pragmatic bedside decision-making,
complementing traditional PRx-based approaches and age-based CPP/MAP targets.
This tool has the potential to enhance precision neurocritical care in
pediatric TBI 38.
Comparison with Existing Literature
Previous
studies in pediatric TBI have largely focused on isolated neuromonitoring
variables, such as ICP, CPP, and PRx, often without considering the dynamic
interplay among cerebral perfusion, systemic hemodynamics, and metabolic
stress. Unlike population-based targets, pICAS provides patient-specific risk
stratification, supporting individualized, physiology-guided perfusion
management. The incremental value of the pICAS lies in its transition from
static measures to functional assessment; while ICP measures static volume and
pressure, the pICAS captures the underlying physiological reserve by
integrating vascular reactivity (PRx) and metabolic stress (lactate). Its
performance (AUC 0.89, internal validation AUC 0.87) aligns with prior adult
and pediatric studies demonstrating the predictive value of multimodal
autoregulation metrics 39,40.
STRENGTHS AND LIMITATIONS
This
study presents several key strengths, including the use of high-quality,
continuous multimodal monitoring in a large pediatric cohort (N=100) and the
integration of cerebral, hemodynamic, and metabolic parameters into a simple,
bedside-applicable score. Furthermore, the findings are supported by robust
internal validation using bootstrap resampling.
Despite
these strengths, certain limitations must be acknowledged, primarily the
retrospective design, which may introduce selection bias. Since the study's
data were collected from tertiary-level centers with advanced monitoring
capabilities, the findings may be skewed toward resource-rich settings,
potentially limiting the applicability of the pICAS to less-equipped centers.
Additionally, referral patterns might have led to the inclusion of patients
with specific profiles, influencing the generalizability of the results.
Furthermore, PbtO₂ monitoring was incomplete in some patients. To address these
limitations, future studies could include broader sampling across hospital
settings to minimize selection bias and better reflect the broader population.
Efforts to ensure more comprehensive monitoring by implementing standardized
protocols across centers could also enhance data completeness. Moreover,
external validation across diverse multicenter pediatric cohorts is essential
to confirm the generalizability of the pICAS score.
Future Directions
Prospective studies are necessary to evaluate real-time pICAS-guided interventions for CPP optimization and vasopressor titration, and to implement digital dashboards for continuous bedside scoring. Moreover, future research should focus on integrating advanced neuromonitoring modalities, such as microdialysis and transcranial Doppler (TCD), to refine risk stratification and improve neurological outcomes in pediatric TBI. Will real-time pICAS feedback alter vasopressor dosing patterns and outcomes? Framing the next trial as a question may inspire collaborative exploration.
Prospective studies are necessary to evaluate real-time pICAS-guided interventions for CPP optimization and vasopressor titration, and to implement digital dashboards for continuous bedside scoring. Moreover, future research should focus on integrating advanced neuromonitoring modalities, such as microdialysis and transcranial Doppler (TCD), to refine risk stratification and improve neurological outcomes in pediatric TBI. Will real-time pICAS feedback alter vasopressor dosing patterns and outcomes? Framing the next trial as a question may inspire collaborative exploration.
CONCLUSIONS
The Pediatric Integrated Cerebral Autoregulation Score
(pICAS) represents a novel, evidence-based bedside tool for early
identification of children with severe traumatic brain injury (TBI) at high risk
of impaired cerebral autoregulation and adverse neurological outcomes. By
integrating multimodal cerebral parameters, including intracranial pressure
(ICP), pressure reactivity index (PRx), deviation from individualized optimal
cerebral perfusion pressure (CPPopt), and brain tissue oxygenation (PbtO₂),
with systemic hemodynamic and metabolic variables such as mean arterial
pressure (MAP) variability, vasopressor dose and duration, and lactate
dynamics, pICAS provides a comprehensive framework reflecting the
multidimensional pathophysiology of autoregulatory failure in pediatric TBI.
Unlike approaches relying solely on isolated metrics or
age-based CPP targets, pICAS contextualizes cerebrovascular reactivity within
systemic physiology, enabling individualized risk stratification and
precision-guided neurocritical care. It supports early identification of
children most likely to benefit from targeted interventions, including
optimized vasopressor titration, CPP management tailored to each child's
autoregulatory capacity, and proactive neuroprotective strategies.
Additionally, pICAS can assist in prioritizing monitoring resources, guiding
escalation of care, and informing real-time decision-making in high-acuity PICU
settings.
Our findings emphasize that impaired autoregulation in
pediatric severe TBI is not a static cerebral phenomenon but a dynamic
interplay of intracranial, systemic, and metabolic factors. By capturing the
cumulative burden across these domains, pICAS moves beyond single-parameter
assessment, offering a holistic, physiologically informed approach to pediatric
TBI management.
While internal validation demonstrates robust predictive
performance and reproducibility, prospective multicenter external validation is
warranted to confirm generalizability across diverse pediatric populations. We
are currently planning a multicenter validation study spanning several
institutions across different geographic regions to thoroughly evaluate the
pICAS tool in varied clinical settings. We invite collaboration from research
centers interested in contributing to this effort, as their involvement would
be invaluable in ensuring the clinical relevance and applicability of the
pICAS. The tool effectively identifies children at high risk of secondary
injury in severe TBI. Future applications may include real-time digital
dashboards, adaptive algorithms to guide individualized CPP targets, and
interventional studies assessing the impact of pICAS-guided management on
long-term neurological outcomes and mortality.
In summary, the pICAS offers a promising, multidimensional
framework for risk stratification in pediatric TBI. While our results
demonstrate its potential utility in identifying patients at risk of secondary
injury, prospective external validation is strictly required before this tool
can be considered a definitive guide for clinical decision-making. Future
research should confirm its incremental value as an actionable bedside metric.
To further the impact of our research, we invite medical professionals and
researchers to validate the pICAS within their own datasets and clinical
environments. By sharing findings and adaptations, the broader community can
collectively refine and optimize this tool, fostering communal progress in
pediatric neurocritical care.
Supplementary
Materials: No supplementary materials are
available for this article.
Author
Contributions: Marlon Carbonell González and
Rosali Santiago Roibal contributed equally as co-first authors to this work.
Marlon Carbonell González: conceptualization, study design, data collection,
statistical analysis, manuscript drafting, and corresponding author responsibilities.
Rosali Santiago Roibal: conceptualization, study design, data collection,
statistical analysis, and manuscript drafting. Deborah Cabrera Rodríguez and
Jorge Luis Ayala Perez: literature review, manuscript revision, interpretation
of clinical findings, and critical review of intellectual content. All authors
have read and approved the final version of the manuscript and agree to be
accountable for all aspects of the work.
Funding: This research received no external funding.
Institutional
Review Board Statement: The authors
are accountable for all aspects of the work in ensuring that questions related
to the accuracy or integrity of any part of the work are appropriately
investigated and resolved. The study was conducted in accordance with the
Declaration of Helsinki and its subsequent amendments. Due to the study's
retrospective nature and the use of anonymized medical records, the ethics
committee waived the requirement for written informed consent.
Informed
Consent Statement: Patient
consent was waived due to the study's retrospective, observational design and
the use of anonymized medical records, in accordance with institutional
requirements and the Declaration of Helsinki.
Data
Availability Statement: The individual
patient data supporting the findings of this study are not publicly available
due to institutional ethical restrictions and the confidentiality of pediatric
patient records.
Acknowledgments: The authors sincerely thank all those involved in patient care and
clinical documentation for their work, which made this study possible.
Conflicts
of Interest: All authors have completed the
ICMJE uniform disclosure form. The authors declare no conflicts of interest.
AI-Assisted Tools Disclosure: The artificial intelligence tool GPAI (https://gpai.app/) was used solely to generate the graphical abstract included in
this study. No artificial intelligence system was used to generate, manipulate,
or analyze experimental data or statistical results. The authors independently
verified all results, analyses, and conclusions, in compliance with the
BioNatura Journal policy: https://bionaturajournal.com/artificial-intelligence--ai-.htmlNo supplementary
materials are available for this article.
REFERENCES
1.
Velle
F, Lewén A, Hånell A, Howells T, Nilsson P, Enblad P. The effects of cerebral
pressure autoregulation status and cerebral perfusion pressure levels on
cerebral metabolism in pediatric traumatic brain injury. Acta Neurochir (Wien).
2024;166(1):190. https://doi.org/10.1007/s00701-024-06085-z
2.
Van Twist E, Robles TB, Formsma B, et al. An open source autoregulation based neuromonitoring algorithm
in pediatric severe traumatic brain injury. Journal of Clinical Monitoring and
Computing (2025) 39:291–299. https://doi.org/10.1007/s10877-024-01248-w
3.
Svedung
Wettervik T, Beqiri E, Hånell A, et al. Intracranial pressure, pressure
reactivity index, cerebral perfusion pressure, and deviation from optimal CPP
in pediatric traumatic brain injury: combined effect on outcome. Childs Nerv
Syst. 2023;39:2459–2466. https://doi.org/10.1007/s00381-023-05982-5
4.
Nelson
LD, et al. Monitoring cerebrovascular reactivity in pediatric traumatic brain
injury: pressure reactivity index versus mean flow index versus cerebral
oximetry index. Neurocrit Care. 2021;37(10):3057–3065. https://doi.org/10.1007/s00381-021-05263-z
5.
Kochanek
PM, Tasker RC, Carney N, et al. Guidelines for the management of pediatric
severe traumatic brain injury. J Neurotrauma. 2019;36:1–60. https://doi.org/10.1097/pcc.0000000000001735
6.
Ducharme
Crevier L. Cerebrovascular pressure reactivity in children with traumatic brain
injury. Pediatr Crit Care Med. 16(8): 2015;p 739-749. https://doi.org/10.1097/pcc.0000000000000471
7.
Brady
KM, et al. Continuous monitoring of cerebrovascular pressure reactivity after
traumatic brain injury in children. Pediatrics. 2009;124:e331–e338. https://doi.org/10.1542/peds.2009-0550
8.
Agrawal
S, et al. STARSHIP: autoregulation status and outcome in severe pediatric head
injury. EClinicalMedicine. 2025;81:103077. https://doi.org/10.1016/j.eclinm.2025.103077
9.
Czosnyka
M, Smielewski P, Kirkpatrick P, et al. Continuous assessment of cerebral
vasomotor reactivity in head injury. Neurosurgery. 1997;41:11–19. https://doi.org/10.1097/00006123-199707000-00005
10.
Steiner LA, Czosnyka M, Smielewski P, et al. Continuous monitoring of cerebrovascular pressure reactivity
allows determination of optimal cerebral perfusion pressure. Crit Care Med.
2002;30:733–738. https://doi.org/10.1097/00003246-200204000-00002
11.
Depreitere
B, et al. Pressure autoregulation monitoring and cerebral perfusion pressure
target recommendations in severe traumatic brain injury. J Neurosurg.
2014;120:1471–1477. https://doi.org/10.3171/2014.3.jns131500
12.
Riemann L, Beqiri E, Smielewski P, et al. Predictive and discriminative power of pressure reactivity
indices in traumatic brain injury. Neurosurgery 87(4):p 655-663,
October 2020.. https://doi.org/10.1093/neuros/nyaa039
13.
Lang
EW, Kasprowicz M, Smielewski P, et al. Short versus long pressure reactivity
index in traumatic brain injury. J Neurosurg. 2015;122:588–596. https://doi.org/10.3171/2014.10.jns14602
14.
Sorrentino
E, et al. Critical thresholds for cerebrovascular reactivity after traumatic
brain injury. Neurocrit Care. 2012;16:258–266. https://doi.org/10.1007/s12028-011-9630-8
15.
Tsigaras ZA, Weeden M, et al. The
pressure reactivity index as a measure of cerebral autoregulation. Crit Care
Resusc. 2023;25:229-236. https://doi.org/10.1016/j.ccrj.2023.10.009
16.
Zeiler
FA, Biasetti J, et al. Continuous cerebrovascular reactivity monitoring after
traumatic brain injury: a review. Br J Anaesth. 2020;124:440–453. https://doi.org/10.1016/j.bja.2019.11.031
17.
Fedriga
M, et al. Cerebral autoregulation in pediatric and neonatal intensive care: a
scoping review. J Intern Med. 2024;295: 1208-1226. https://doi.org/10.1177/0271678x241261944
18.
Svedung
Wettervik T, et al. Cerebral pressure autoregulation in brain injury: a review.
World Neurosurg. 2022;161: 118-131. https://doi.org/10.1016/j.wneu.2021.11.027
1.Carney N, Totten AM, O'Reilly C, et al. Guidelines for the
management of severe traumatic brain injury, 4th edition. Neurosurgery. 2017; 1;80(1):6-15. https://doi.org/10.1227/NEU.0000000000001432
20. Brain Trauma Foundation, et al. Guidelines for the management of severe
traumatic brain injury. J Neurotrauma. 2007;24(Suppl 1):S1–S106. https://doi.org/10.1089/neu.2007.9996
21.
Steyerberg
EW, et al. Predicting outcome after traumatic brain injury: development and
validation of prognostic scores. PLoS Med. 2008;5:e165. https://doi.org/10.1371/journal.pmed.0050165
22.
Aries
MJH, Czosnyka M, et al. Continuous determination of optimal cerebral perfusion
pressure in traumatic brain injury. Crit Care Med. 2012;40:2456–2463. https://doi.org/10.1097/CCM.0b013e3182514eb6
23.
Beqiri E, Smielewski P, et al. COGiTATE study: feasibility of individualized cerebral
perfusion pressure management in traumatic brain injury. BMJ Open.
2019;9:e030727. https://doi.org/10.1136/bmjopen-2019-030727
24.
Beqiri
E, et al. Lower limit of cerebrovascular reactivity as a personalized cerebral
perfusion pressure target in traumatic brain injury. Crit Care. 2023;27:194. https://doi.org/10.1186/s13054-023-04485-8
25.
Bögli
SY, et al. Dynamic versus fixed cerebral perfusion pressure targets in
pediatric traumatic brain injury: a STARSHIP analysis. EClinicalMedicine.
2025;86:103370. https://doi.org/10.1016/j.eclinm.2025.103370
26.
Smith
CA, et al. Exploration of cerebral perfusion pressure targets in pediatric
traumatic brain injury. EClinicalMedicine. 2025;29:207. https://doi.org/10.1186/s13054-025-05458-9
27.
Gritti
P, et al. Cerebral autoregulation in traumatic brain injury across age groups. Crit
Care 28, 33 (2024). https://doi.org/10.1186/s13054-024-04814-5
28.
Bayir H, Kagan VE, et al. Assessment
of antioxidant reserves and oxidative stress in cerebrospinal fluid after
severe traumatic brain injury in infants and children. Pediatr Res.
2002;51:571–578. https://doi.org/10.1203/00006450-200205000-00005
29.
Evensen
KB, Eide PK. Limitations and potential improvements in intracranial pressure
measurement. Fluids Barriers CNS. 2020;17:15. https://doi.org/10.1186/s12987-020-00195-3
30.
Young
AMH, Guilfoyle MR, et al. Application of adult traumatic brain injury models in
a pediatric cohort. J Neurosurg Pediatr. 2016;17:558–564. https://doi.org/10.3171/2016.5.PEDS15427
31.
Dewan
MC, Mummareddy N, et al. Epidemiology of global pediatric traumatic brain
injury: a qualitative review. World Neurosurg. 2016;91:497–509. https://doi.org/10.1016/j.wneu.2016.03.045
32.
Kochanek
PM, Tasker RC, et al. Clinical recommendations for pediatric severe traumatic
brain injury. Pediatr Crit Care Med. 2019;20:269–279. https://doi.org/10.1097/pcc.0000000000001737
33.
Mikkonen
ED, Skrifvars MB, et al. Validation of prognostic models in pediatric traumatic
brain injury. J Neurosurg Pediatr. 2019;23:203–210. https://doi.org/10.3171/2019.4.PEDS1983
34.
Oddo
M, Taccone FS, et al. Recommendations for multimodal monitoring in
neurointensive care. Curr Opin Crit Care. 2015;21:135–143. https://doi.org/10.1097/MCC.0000000000000179
35.
Depreitere
B, et al. Minute by minute pressure reactivity and cerebral perfusion pressure
monitoring in severe traumatic brain injury. J Neurosurg. 2014;120:1471–1477. https://doi.org/10.3171/2014.3.jns131500
36.
Stefan
Y, et al. Association between EEG metrics and continuous cerebrovascular
autoregulation assessment: a scoping review. Br J Anaesth. 2024;133(3):550–564.
https://doi.org/10.1016/j.bja.2024.03.021
37.
Riemann
L, et al. Low-resolution pressure-reactivity index and its derived optimal
cerebral perfusion pressure in adult patients with traumatic brain injury: a
CENTER-TBI study. Crit Care. 2020;24:266. https://doi.org/10.1186/s13054-020-02974-8
38.
Zeiler
FA, et al. Cerebrovascular reactivity monitoring: adult and pediatric
relevance. Br J Anaesth. 2020;124:440–453. https://doi.org/10.1016/j.bja.2019.11.031
39.
Czosnyka
M, Pickard JD. Cerebral autoregulation following head injury. J Neurosurg.
2001;95:756–763. https://doi.org/10.3171/jns.2001.95.5.0756
40.
Donnelly
J, et al. Individualizing thresholds of cerebral perfusion pressure using
estimated limits of autoregulation. Crit Care Med. 2017;45(9):1464–1471. https://doi.org/10.1097/CCM.0000000000002575
Received: February 2,
2026 / Accepted:
April 5, 2026 / Published (Online First): April
17, 2026 / Issue Date: June 15, 2026
(Europe/Madrid)
Citation: Carbonell González M, Santiago Roibal R, Cabrera Rodríguez D, Ayala Pérez
JL. Integrated Cerebral Autoregulation Score for Early Risk
Stratification in Severe TBI. BioNatura Journal: Ibero-American Journal of
Biotechnology and Life Sciences. 2026;3(2):3.
https://doi.org/10.70099/BJ/2026.03.02.3
Correspondence should be addressed to:
marloncarbonell95@gmail.com
Peer Review Information: BioNatura
Journal thanks the anonymous reviewers for their valuable
contribution to the peer-review process. Regional peer-review coordination was
conducted under the BioNatura Institutional Publishing Consortium (BIPC).
Reviewer selection and assignment were supported via: https://www.reviewercredits.com/
Digital Preservation and Repository:
This journal is managed through the Open Journal Systems (OJS) platform. To
ensure long-term access, we use the PKP Preservation Network (PKP PN) to
digitally preserve all published volumes in a decentralized, secure archive.
Furthermore, our repository is integrated with LOCKSS and CLOCKSS, allowing
international library networks to create permanent archives for long-term
survival.
Publisher Information:
Published by Clinical Biotec S.L. (Madrid, Spain) as the publisher of record
under the BioNatura Institutional Publishing Consortium (BIPC). Places of
publication: Madrid (Spain); Tegucigalpa (Honduras); Panama City (Panama).
Online ISSN: 3020-7886.