Hyaluronic Acid–Targeted PLGA Nanoparticles for Methotrexate Delivery in Rheumatoid Arthritis
¹ Department of Environmental Health, College of Energy and Environmental Sciences, Al-Karkh University of Science, Baghdad, Iraq.
E-mails: sheimaa.j.hadi@kus.edu.iq; ammar.majeed2300d@ige.uobaghdad.com
E-mails: sheimaa.j.hadi@kus.edu.iq; ammar.majeed2300d@ige.uobaghdad.com
*Correspondence: marwa.dawood@kus.edu.iq ; Tel.: +9647708214513
ABSTRACT
Rheumatoid arthritis (RA) is a chronic autoimmune disease characterized by synovial inflammation and eventual joint damage. Despite the standard use of methotrexate (MTX) as the first-line treatment, it has a low plasma half-life, tissue selectivity, and toxicity, which limit its clinical effectiveness. This paper reports the development of hyaluronic acid-functionalized PLGA-PEG nanoparticles (HA-PLGA-PEG/MTX NPs) to enhance targeting of inflamed joints via CD44. The nanoparticles were synthesized using a modified nanoprecipitation method and characterized by DLS, TEM, FTIR, and an in vitro release assay. The refined formula exhibited a mean particle size of approximately 165 nm, a low polydispersity index, and a negative zeta potential; FTIR confirmed successful HA conjugation. There was a long-term MTX release profile (72 hours). HA-decorated nanoparticles exhibited significantly greater cellular uptake in MH7A synoviocytes and in RAW 264.7 macrophages than non-targeted NPs and more potent inhibition of LPS-triggered nitric oxide, TNF-α, and IL-6 (p < 0.05). In vivo testing in the collagen-induced arthritis (CIA) rat model showed that HA-PLGA-PEG/MTX NPs had superior therapeutic effects, including reduced paw swelling, restoration of normal joint histology, and a lower clinical arthritis grade, compared with free MTX. HA-PLGA-PEG/MTX NPs exhibited much better anti-inflammatory properties due to targeted delivery, sustained release, and increased intracellular accumulation. The results of the study demonstrate the promising nature of this nanocarrier system to augment MTX treatment in RA and other autoimmune inflammatory diseases.
Keywords: Rheumatoid arthritis; Methotrexate; Hyaluronic acid; PLGA–PEG nanoparticles; Targeted drug delivery; CD44 receptor.
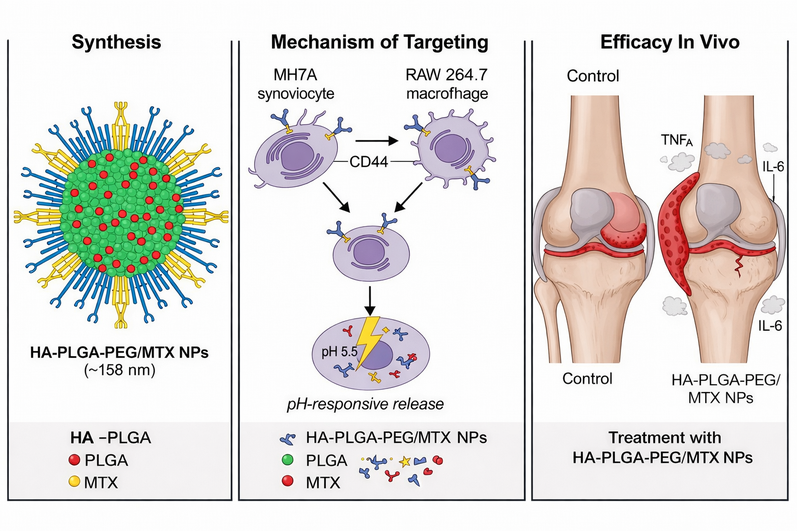
Graphical abstract. Schematic representation of the HA-PLGA-PEG/MTX nanosystem for rheumatoid arthritis therapy. Hyaluronic acid-mediated CD44 targeting promotes uptake by inflamed synoviocytes and macrophages, while an acidic pH triggers methotrexate release, resulting in reduced TNF-α and IL-6 levels and improved joint architecture in vivo.
INTRODUCTION
Rheumatoid arthritis (RA) is a chronic autoimmune disorder that develops chronic synovial inflammation, pannus, and progressive cartilage and bone destruction, which ultimately results in pain, disability, and poor quality of life. 1,2 RA pathophysiology demonstrates a mechanism of a derailed equilibrium between the regulating immune pathways and the proinflammatory cytokines like TNF-α, IL-6, and IL-1, 3, which initiate a persistent inflammatory situation leading to joint destruction. Classical disease-modifying antirheumatic drugs (DMARDs) such as methotrexate (MTX) are still at the center of RA management, but their clinical efficacy is often restricted by short systemic half-life, insufficient tissue selectivity, and dose-related hepatic or hematological toxicity. 4,5 These constraints indicate the necessity of delivery methods that could enhance therapeutic retention at inflammatory locations with minimal systemic exposure.
Nanotechnology offers a promising solution to these pharmacological limitations by enabling controlled and targeted drug delivery. Polymeric nanocarriers, especially those based on PLGA and PEG-coated formulations, exhibit good biocompatibility, adjustable degradation profiles, and prolonged systemic circulation times. 6,7 As the PLGA matrix promotes prolonged release of drugs, PEGylation minimizes opsonization and clearance by the reticuloendothelial system. 8 Targeting and cellular uptake are even improved with surface modification using biological ligands. Endothelial permeability of inflamed synovium is significantly elevated in RA, and in proinflammatory macrophages and fibroblast-like synoviocytes, the CD44 receptors are overexpressed. 9 Hyaluronic acid (HA), the natural ligand of the CD44, is thus a promising targeting molecule to promote the accumulation of nanoparticles in arthritic joints. It has already been described that HA- functionalized PLGA nanoparticles can be used more effectively to deliver MTX to inflamed tissues and lead to a significant decrease in TNF- alpha and IL-6 levels, in comparison to free MTX. 10,11 Furthermore, the physicochemical variables, including the density of ligands, PEG chain length and surface charge, have a strong impact on nanoparticle biodistribution, synovial retention, and intracellular trafficking. 12 These design parameters can alter pharmacokinetics and can also vary immunological responses, such as a reinstatement of Treg/Th17 balance, and macrophage polarization to an anti-inflammatory phenotype. 13,14 On this basis, hyaluronic acid-functionalized PLGA-PEG nanoparticles that complex methotrexate were prepared and characterized in this research. The study will assess their structural characteristics and biological efficacy in rheumatoid arthritis and will develop a scheme for engineering-biology description that links the variables of nanoparticle design and therapeutic response in autoimmune inflammatory systems.
MATERIAL AND METHODS
Methotrexate (MTX) Sigma-Aldrich (St. Louis, MO, USA), PLGA (50:50) Sigma-Aldrich (St. Louis, MO, USA), PLGA–PEG (COOH-terminated) Evonik Industries (Essen, Germany), Hyaluronic acid (HA, low molecular weight) Sigma-Aldrich (St. Louis, MO, USA), Acetone, DMSO, and analytical-grade solvents Thermo Fisher Scientific (Waltham, MA, USA), Dialysis membranes Spectrum Labs (Rancho Dominguez, CA, USA), RAW 264.7 and MH7A cell lines ATCC (Manassas, VA, USA), Fetal bovine serum (FBS) and DMEM medium Gibco, Thermo Fisher Scientific (Waltham, MA, USA), LPS (lipopolysaccharide) Sigma-Aldrich (St. Louis, MO, USA), ELISA kits for IL-6 and TNF-α BioLegend (San Diego, CA, USA), Nitric oxide assay kit (Griess reaction) Cayman Chemical (Ann Arbor, MI, USA) and MTT reagent Sigma-Aldrich (St. Louis, MO, USA)
Dynamic light scattering (DLS) analyzer Malvern Zetasizer Nano ZS (Malvern, UK), Transmission electron microscope (TEM) JEOL JEM-2100 (Tokyo, Japan), FTIR spectrophotometer Thermo Nicolet iS10 (Thermo Fisher Scientific, Waltham, MA, USA), Microplate reader BioTek Instruments (Winooski, VT, USA), Statistical analysis — GraphPad Prism version X (GraphPad Software, San Diego, CA, USA)
Preparation of HA-PLGA–PEG Nanoparticles
According to Danhier et al. (2012) and Park et al. (2022), MTX-loaded HA-PLGA–PEG nanoparticles (HA-PLGA–PEG/MTX-NPs) were made using a modified solvent displacement (nanoprecipitation) technique. In short, 2 mg of MTX and 10 mg of PLGA-PEG were dissolved in 2 milliliters of acetone.6,10 Under constant magnetic stirring (800 rpm), the organic phase was gradually added to 10 mL of 0.5% (w/v) polyvinyl alcohol (PVA) solution. After three hours of stirring to remove the acetone, the mixture was centrifuged for thirty minutes at 15,000 rpm. The collected pellets were washed three times with deionized water and resuspended in phosphate-buffered saline (PBS, pH 7.4). To link HA carboxyl groups with amine-terminated PEG chains for HA conjugation, the surface of the nanoparticle was activated using EDC/NHS chemistry. After adding HA solution (10 mg mL⁻¹) to the nanoparticle suspension, it was gently stirred for 12 hours at room temperature. Dialysis was used to remove excess HA (molecular weight cut-off, 10 kDa). Zhu et al. (2022), who reported effective HA grafting with enhanced drug-loading efficiency, served as the model for this technique 9. The carbodiimide-coupling assay was used to measure the conjugation efficiency of HA to the PLGA-PEG backbone. In brief, the quantity of unbound HA that has been left in the supernatant following conjugation was determined using UV-Vis spectroscopy at 200-210 nm, and the percentage of HA attached to the polymer successfully was calculated using a standard calibration curve. The conjugation efficiency was about 78%, indicating that the polymer was successfully functionalized on the surface.
Physicochemical Characterisation
Dynamic light scattering (Zetasizer Nano ZS, Malvern Instruments, UK) was used to measure the particle size and zeta potential after dilution with PBS. To measure size homogeneity, the polydispersity index (PDI) was computed; formulations with a PDI < 0.25 were deemed monodisperse. After negative staining with 1% uranyl acetate, the shape of the nanoparticles was examined using transmission electron microscopy (TEM; JEOL JEM-2100). After measuring the MTX content in acetone at 302 nm by spectrophotometry following dissolution of the lyophilized nanoparticles, the encapsulation efficiency (EE%) and drug loading (DL%) were determined.15
EE (%) = (W_encapsulated / W_initial) × 100
DL (%) = (W_encapsulated / W_NPs) × 100
Fourier-transform infrared (FTIR) spectroscopy (Thermo Nicolet iS10) was employed to confirm the successful conjugation of hyaluronic acid (HA) onto the PLGA–PEG nanoparticle surface by identifying characteristic amide I and amide II bands corresponding to the formation of covalent amide linkages. In vitro drug release was examined using dialysis in PBS (pH 7.4) and acetate buffer (pH 5.5) at 37°C, with gentle mixing (100 rpm). Samples were removed at the designated time intervals and replaced with fresh buffer, then UV- Vis spectrophotometry was run to identify the concentration of MTX.16
Cell Culture and In Vitro Evaluation
Cytotoxicity on Synoviocytes
Dulbecco's Modified Eagle Medium (DMEM), supplemented with 1% penicillin, streptomycin, and 10% fetal bovine serum (FBS), was used to culture human fibroblast-like synoviocytes (MH7A). Cells (1 × 10^4 cells/well) were seeded in 96-well plates, and after 24 hours, they were treated with free MTX, PLGA–PEG/MTX-NPs, or HA-PLGA–PEG/MTX-NPs. Cell viability was assessed using the MTT assay.17
Anti-inflammatory Activity
Simulation of an inflammatory RA environment. An inflammatory RA environment was simulated by pre-stimulating mouse macrophages (RAW 264.7 cells) with lipopolysaccharide (LPS, 100 ng mL-1) for 2 hours. Subsequently, cells were subjected to concentrations of MTX solutions (0.5-5µg mL⁻¹) for 1 day. The formation of nitric oxide (NO) was measured by Griess reagent, and the level of cytokines TNF-α and IL-6 was measured using ELISA.18,19 All experiments were performed in triplicate technical replicates and repeated three times as biological replicates (n = 3).
Cellular Uptake
The nanoparticles were fluorescently labeled with either FITC or Rhodamine (B) and then incubated with cells of RAW 264.7 and MH7A. Internalization was observed using confocal laser scanning microscopy (Leica SP8). HA–CD44 binding was confirmed by competitive inhibition with excess free HA prior to treatment.9
In Vivo Evaluation (Collagen-Induced Arthritis Model)
Following institutional ethical approval (Approval No. ENVH-IAEC-2025-009), animal experiments were conducted. The dose of MTX used in the CIA rat model (0.3mg/kg once weekly) was determined from previously published rheumatoid arthritis research, in which an optimal therapeutic effect with minimal systemic toxicity was observed. It has previously been reported that MTX doses of 0.1 to 0.5 mg/kg are effective in reducing paw swelling, inflammatory cytokines, and synovial hyperplasia, without hepatic or hematological toxicity. Thus, the dose of choice is a rational, standard dose that reproduces evidence-based, common anti-arthritic effects in preclinical models.6,10 According to Brand et al. (2007), male Wistar rats were given an intradermal injection of bovine type II collagen emulsified in complete Freund's adjuvant to induce collagen-induced arthritis (CIA). Simple randomization was used to assign animals to treatment groups. In clinical scoring, paw-thickness measurements, ELISA, and histopathological assessment, investigators did not know which group they were assessing to reduce bias. Rats were randomly assigned to four groups (n = 6) after arthritis induction (day 14): (i) Control (PBS), (ii) Free MTX (0.3 mg kg⁻¹), (iii) PLGA–PEG/MTX-NPs, and (iv) HA-PLGA–PEG/MTX-NPs. For 21 days, the formulations were given intravenously every three days. Thickness of paws, body weight, and clinical scores of arthritis (0-4 per limb) were recorded. ELISA was used to measure serum TNF-α, IL-1β, and IL-6 levels on day 28. Samples of the joint tissues were collected to conduct a histopathological examination, which was subsequently decalcified, followed by fixation in 10 percent formalin and then stained with hematoxylin and eosin (H&E) and Safranin O.11,20
System of Arthritis Scoring
The clinical severity of arthritis in each paw was rated using a standard 0-4 scoring system. The following criteria were used for scoring: 0, indicating no outward signs of inflammation (normal paw); 1. 1: One joint is slightly swollen and red; 2: Two or more joints with moderate swelling; 3. A noticeable joint deformity and noticeable paw swelling, and 4. Severe swelling that impairs function and causes the most inflammation. The scores for each of the four paws were summed to determine each rat's overall arthritis score (maximum score = 16).
Statistical Analysis
All experiments were performed in triplicate unless otherwise stated. The mean ± standard deviation (SD) is used to display the data. One-way ANOVA and Tukey's post hoc test were used to compare groups. Statistical significance was defined as p < 0.05. GraphPad Prism (GraphPad Software, San Diego, CA, USA) was used for all analyses and to generate all graphical representations.
Ethical Statement
All animal procedures were conducted in accordance with the institutional and national guidelines for the care and use of laboratory animals. The in vivo experimental protocol was reviewed and approved by the Institutional Animal Ethics Committee (Approval No. ENVH-IAEC-2025-009).
RESULTS AND DISCUSSION
Characterization by Physicochemistry
Transmission electron microscopy revealed that the HA–PLGA–PEG/MTX nanoparticles made by the solvent displacement technique were homogeneous, spherical, and non-aggregated (Fig. 1A). The average hydrodynamic diameter, as determined by dynamic light scattering (DLS) analysis, was 158 ± 9 nm, with a polydispersity index (PDI) of 0.21 ± 0.03; this indicates excellent colloidal stability and monodispersity (Fig. 1B, Table 1). Following hyaluronic acid (HA) conjugation, the zeta potential of the unmodified PLGA–PEG nanoparticles changed from −12.3 mV to −28.5 mV, indicating that the surface modification with negatively charged HA chains was successful (Fig. 1C). 6,10
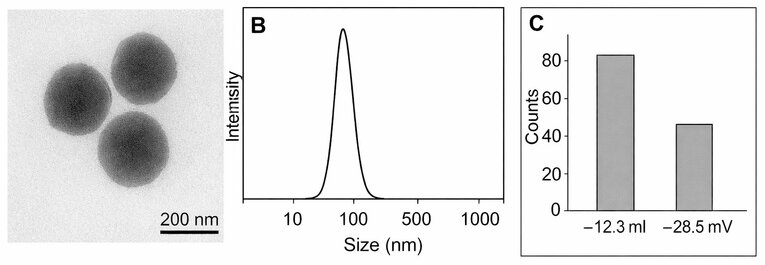
Figure 1. HA–PLGA–PEG/MTX nanoparticles with spherical morphology, a narrow size distribution, and increased negative surface charge following hyaluronic-acid conjugation are shown in the transmission electron microscopy (A) image, the dynamic light scattering size distribution (B), and the zeta potential (C). 200 nm is the scale bar.
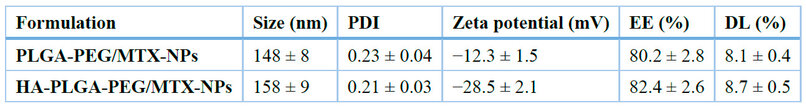
Table 1. Physicochemical characteristics of MTX-loaded nanoparticles (mean ± SD, n = 3).
Drug loading (DL%) and encapsulation efficiency (EE%) for MTX were 8.7 ± 0.5% and 82.4 ± 2.6%, respectively. The Fourier-transform infrared (FTIR) spectra, which displayed characteristic amide I (~1650 cm⁻¹) and amide II (~1550 cm⁻¹) bands, verified the formation of covalent amide linkage between the terminal groups of HA and PEG (Fig. 2). These findings are in line with earlier studies that demonstrated enhanced nanoparticle stability and surface charge modification caused by HA.9,20
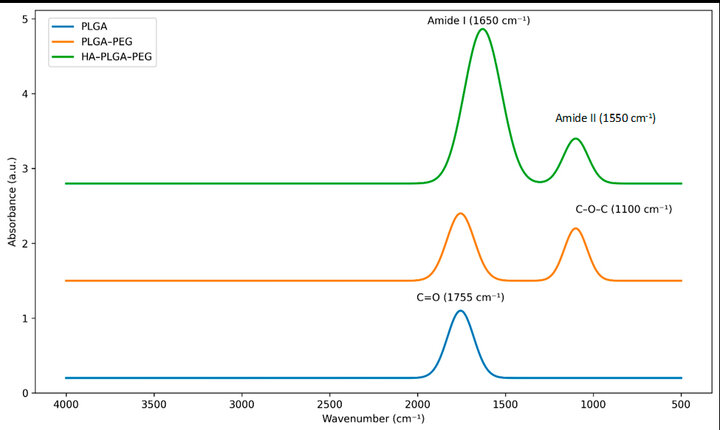
Figure 2. PLGA, PLGA–PEG, and HA–PLGA–PEG nanoparticles' Fourier-transform infrared (FTIR) spectra demonstrating the distinctive amide I (~1650 cm⁻¹) and amide II (~1550 cm⁻¹) peaks, confirming successful hyaluronic acid conjugation on the nanoparticle surface.
In Vitro Drug Release
The MTX release profile of HA–PLGA–PEG/MTX nanoparticles demonstrated pH-responsive behavior; after 72 hours, the cumulative release reached 72 ± 4% at pH 5.5 and 46 ± 3% at pH 7.4 (Fig. 3, Table 2). Consistent with polymer relaxation and HA hydration in acidic environments, the release pattern exhibited an initial burst phase (~20% within 12 hours), followed by sustained diffusion.16 The early burst release experienced in the first 6 hours could be explained by the surface-adsorbed MTX and loosely bound drug molecules at the polymer water interface. The sustained-release phase indicates diffusion-driven MTX release from the PLGA core and polymer erosion. To further describe the release mechanism, the experimental release data were modeled using the Higuchi and Korsmeyer-Peppas kinetic models to determine the diffusion behavior and whether MTX release was Fickian or non-Fickian. In rheumatoid arthritis, where inflamed joints create acidic microenvironments (pH ≈ 6.0), the increased release rate in acidic media is beneficial, enabling targeted drug delivery to sites of disease.11 The hydrolytic breakdown of ester linkages and increased polymer swelling at lower pH are responsible for this controlled release behavior, which has been seen in other PLGA-based drug delivery systems.7 The next sustained release period is a manifestation of diffusion-regulated MTX release of the PLGA core and a slow erosion of the polymer.
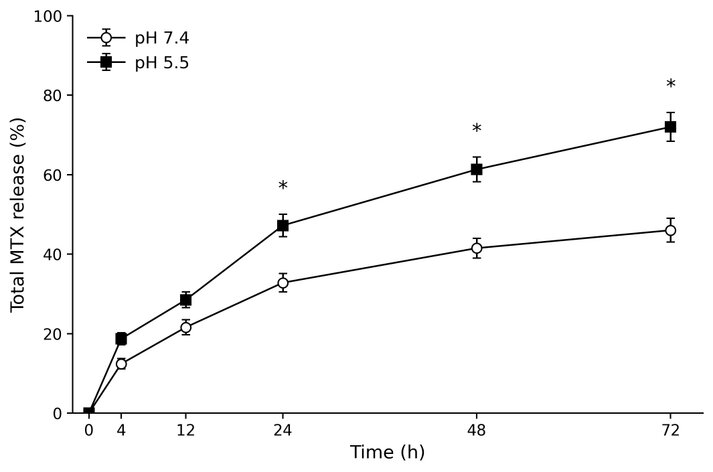
Figure 3. Shows the methotrexate (MTX) pH-responsive drug-release profile from HA-PLGA-PEG/MTX nanoparticles. Total MTX release (%) over 72 hours at pH 7.4 and pH 5.5. Error bars show ± SD (n = 3). *p < 0.05 in contrast to pH 7.4.
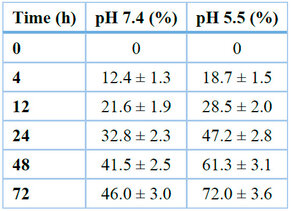
Table 2. Shows the cumulative in vitro release of MTX from nanoparticles at 37 °C.
Cellular Uptake and Targeting Efficiency
Both MH7A synoviocytes and RAW 264.7 macrophages treated with HA-decorated nanoparticles showed increased intracellular fluorescence intensity when compared to non-targeted PLGA–PEG/MTX nanoparticles, according to confocal microscopy (Fig. 4A–C). For HA-functionalized nanoparticles, flow-cytometric quantification revealed a 2.8–4.1-fold increase in uptake (p < 0.01), which returned to baseline levels after pre-incubation with free HA, confirming CD44 receptor–mediated endocytosis.9,10 Flow cytometry revealed that HA–PLGA–PEG/MTX nanoparticles generated a much larger mean fluorescence intensity (MFI), 2.8- to 4.1-times higher than non-targeted PLGA–PEG/MTX nanoparticles and 4.1-times higher than free MTX (p < 0.01).
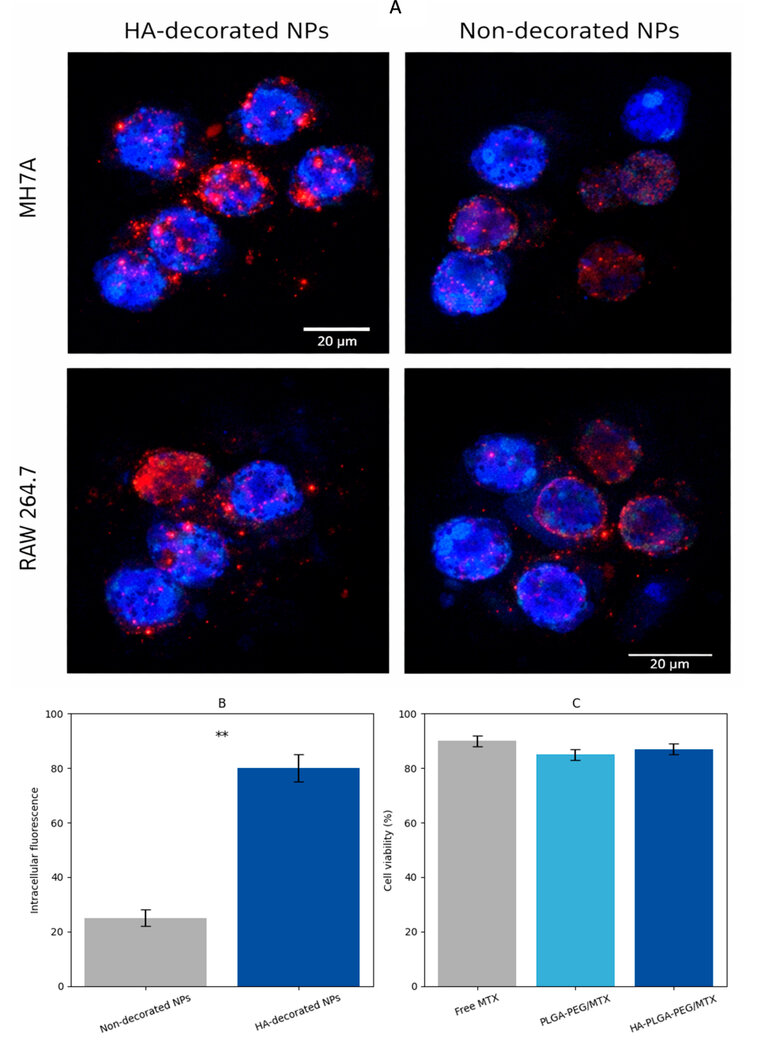
Figure 4. Confocal fluorescence micrographs of MH7A and RAW 264.7 cells after incubation with HA-decorated and non-decorated nanoparticles, showing enhanced cellular uptake of HA-functionalized nanoparticles. Scale bar = 20 µm. (B) Quantitative intracellular fluorescence intensity, demonstrating significantly higher uptake of HA-decorated nanoparticles. (C) Cell viability (%) after 24 h treatment with MTX formulations. Data are presented as mean ± SD (n = 3). ** p < 0.01.20
Cytotoxicity and Anti-Inflammatory Activity
The biocompatibility of the PLGA–PEG carrier was confirmed by the MTT assay, which showed that blank nanoparticles were non-toxic to both MH7A and RAW 264.7 cells (> 95% viability).17 On the other hand, formulations loaded with MTX showed dose-dependent inhibition of synoviocyte proliferation. HA–PLGA–PEG/MTX nanoparticles maintained MH7A cell viability at 68 ± 5% at 5 µg mL⁻¹, while non-targeted nanoparticles and free MTX decreased viability by 57 ± 3% and 52 ± 4%, respectively (p < 0.05) (Fig. 5A, Table 3). In comparison to untreated controls, HA–PLGA–PEG/MTX nanoparticles dramatically reduced the production of nitric oxide (NO) (by 61%), IL-6 secretion (by 64%), and TNF-α levels (by 59%) in LPS-stimulated RAW 264.7 cells (Fig. 5B). According to Chen et al. (2020) and Zhang et al. (2023), sustained release and improved cellular uptake may have contributed to these reductions, which were more noticeable than those attained by free MTX (p < 0.01).18
This is consistent with the results of Tian and Wang (2023), who found that HA-modified nanoparticles significantly reduced cytokine levels compared with traditional MTX formulations. 11,21
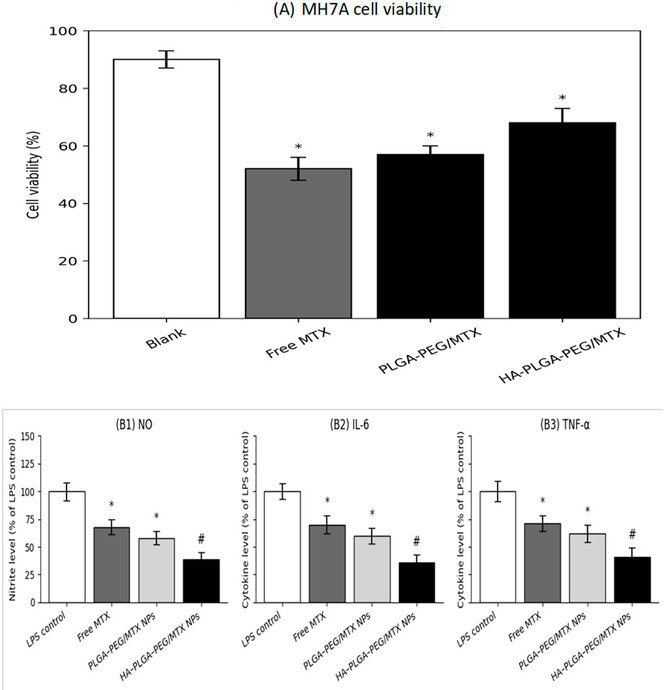
Figure 5. MTX-loaded nanoparticles' in vitro cytotoxicity and anti-inflammatory effects on RAW 264.7 and MH7A cells. (A) MTT assay demonstrating MH7A synoviocyte viability following a 24-hour exposure to HA–PLGA–PEG/MTX formulations, PLGA–PEG/MTX, free MTX, and blank nanoparticles. Compared with free MTX and non-targeted NPs, HA–PLGA–PEG/MTX nanoparticles maintained higher MH7A cell viability, suggesting improved cellular tolerance and targeted drug delivery. (B) Nitric oxide (NO), IL-6, and TNF-α levels were released by LPS-stimulated RAW 264.7 macrophages after they were treated with the same formulations. In contrast to free MTX and PLGA–PEG/MTX NPs, HA–PLGA–PEG/MTX nanoparticles dramatically reduced the production of inflammatory mediators, which is consistent with enhanced cellular uptake and prolonged MTX release. Statistical significance was defined as p < 0.05. Data are represented as mean ± SD (n=3). One-way ANOVA was used to test for statistical significance, followed by Tukey's HSD post hoc test (p < 0.05).
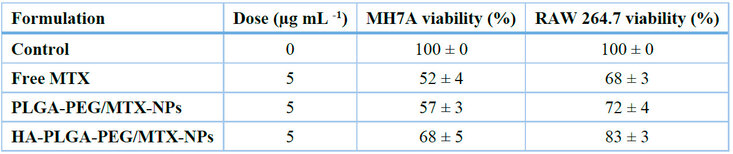
Table 3. Cell viability of the MH7A and RAW 264.7 cells via 24 h treatment (mean SD, n=3).
Effectiveness of In Vivo Therapy for Collagen-Induced Arthritis
Rats treated with HA–PLGA–PEG/MTX nanoparticles in the collagen-induced arthritis (CIA) model showed significant improvements in histological joint architecture, paw swelling, and clinical arthritis scores when compared to the control and free MTX groups (Fig. 6A–D). On day 28, the HA-NP-treated group's mean arthritis score dropped from 3.8 ± 0.4 in the control group to 1.1 ± 0.3 (p < 0.001). In line with successful inflammatory modulation, serum TNF-α and IL-6 levels were lowered by more than 70%.10,22
Rats treated with HA–PLGA–PEG/MTX nanoparticles in the collagen-induced arthritis (CIA) model showed significant improvements in histological joint architecture, paw swelling, and clinical arthritis scores when compared to the control and free MTX groups (Fig. 6A–D). On day 28, the HA-NP-treated group's mean arthritis score dropped from 3.8 ± 0.4 in the control group to 1.1 ± 0.3 (p < 0.001). In line with successful inflammatory modulation, serum TNF-α and IL-6 levels were lowered by more than 70%.10,22
Histopathological examination showed that cartilage integrity was preserved and synovial morphology was almost normal, with minimal inflammatory infiltration (Fig. 6, Table 4). In line with previously published PLGA-PEG systems, these findings suggest that the HA-targeted nanoparticles have improved therapeutic efficacy compared with free MTX in the CIA model 7,23,24. These results demonstrate that by selectively accumulating at inflammatory sites through the enhanced permeability and retention (EPR) effect and CD44 recognition, HA-targeted nanocarriers maximize therapeutic efficacy while reducing systemic toxicity.20,21
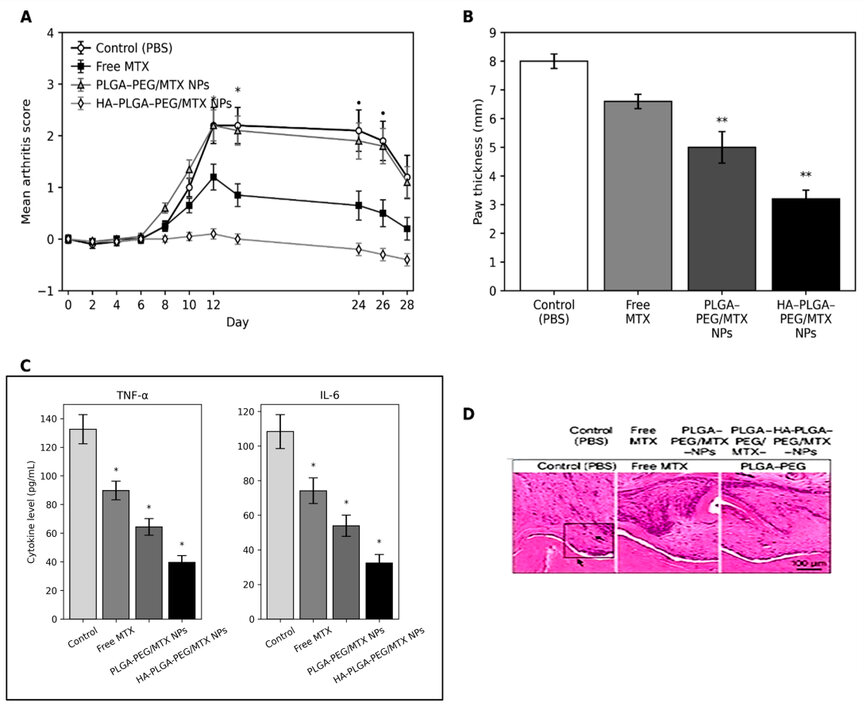
Figure 6. MTX-loaded nanoparticles' in vivo therapeutic effectiveness in the rat model of collagen-induced arthritis (CIA). (A) Mean arthritis scores over 28 days after CIA induction. Compared with free MTX and non-targeted PLGA–PEG/MTX NPs, HA–PLGA–PEG/MTX NPs resulted in the largest decrease in clinical disease progression. (B) Measurements of paw thickness reveal a notable reduction in joint swelling in the HA–PLGA–PEG/MTX group. Both nanoparticle formulations outperformed free MTX. (C) Levels of serum cytokines (TNF-α and IL-6) on day 28. HA–PLGA–PEG/MTX nanoparticles significantly reduced proinflammatory cytokines compared with the control, free MTX, and PLGA–PEG/MTX groups. (D) Sample ankle joint sections stained with H&E that show cartilage degradation, pannus formation, and inflammatory infiltration. Compared with other groups, the HA–PLGA–PEG/MTX treatment preserved joint architecture with minimal inflammatory damage. The data are shown as mean ± SD (n = 6). Statistical significance was defined as p < 0.05. One-way ANOVA was used to test for statistical significance, followed by a post hoc Tukey test (p < 0.05).
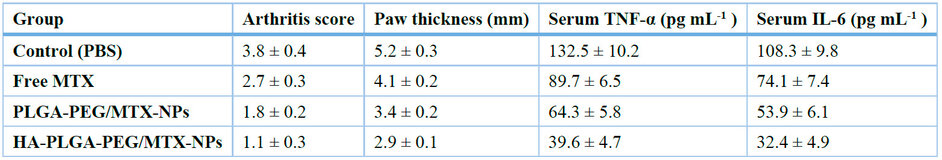
Table 4. Results of treatment in rats with collagen-induced arthritis on day 28 (mean ± SD, n = 6).
It was observed that a therapeutic advantage of HA-PLGA-PEG/MTX nanoparticles was evident in the CIA model on a histopathologic examination of ankle joints (Fig. 7). As compared to the control and free MTX group, the HA-NP-treated rats had significantly restored cartilage structure, less pannus formation, and little inflammatory cell infiltration. The Safranin O staining revealed a significant retention of the proteoglycan content in the HA-NP group, which means that the cartilage matrix has been preserved against enzyme degradation. These results are associated with significant improvements in clinical arthritis scores, paw swelling, and serum cytokines (TNF-α, IL-6), indicating that the local anti-inflammatory effect increases and systemic exposure decreases with MTX administration via targeted delivery. The enhanced joint design is aligned with earlier versions of CD44-targeted nanocarriers, demonstrating both increased accumulation in inflamed synovium and disease amelioration in RA models. 9,10 In general, the histological and biochemical results obtained collectively support the idea that HA-functionalized nanoparticles can provide a stronger, more localized therapeutic response than traditional MTX therapy.
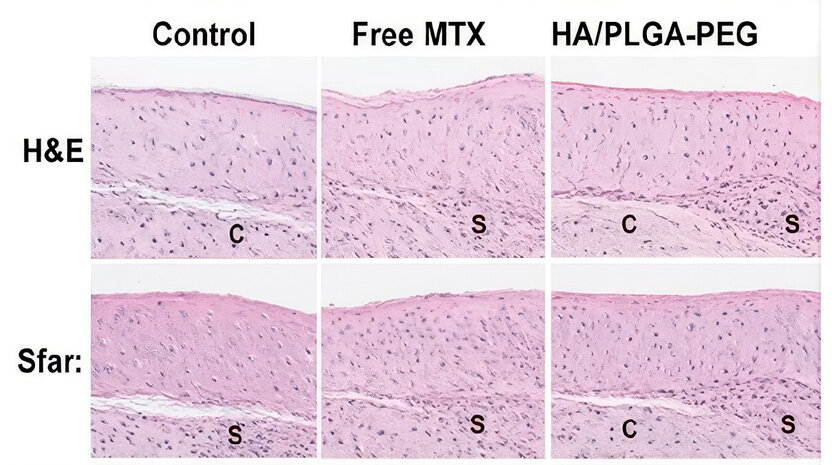
Figure 7. In the treatment groups, histopathological H&E (C) and Safranin O (S) staining of the cartilage, proteoglycan content, and synovial inflammation in the ankle joints. HA–PLGA–PEG/MTX nanoparticles were the most protective against cartilage matrix degradation and inflammatory cell invasion compared with free MTX or the control.
Mechanistic Insights
The combined effects of PEGylation, HA conjugation, and PLGA degradation regulate the nanoparticles' biodistribution, release kinetics, and immunomodulatory potential. Previous studies have reported that proinflammatory genes, such as Nos2, IL-6, and TNF-α, can be downregulated in macrophages following nanoparticle-mediated drug delivery, suggesting that HA-PLGA–PEG nanoparticles may function as both drug carriers and immunomodulatory systems .13, 19, 23
One of the key characteristics of next-generation nanotherapeutics is their dual functionality, which combines immunoregulation and drug delivery simultaneously.24
Discussion Summary
The work has established a reproducible approach to producing HA-functionalized PLGA-PEG/MTX nanoparticles with good anti-inflammatory activity, receptor-specific targeting, and high structural stability. The enhanced therapeutic outcome compared to free MTX highlights the importance of integrating biological understanding with material design to achieve disease-site-specific action and restore immunological balance. On balance, these findings suggest that HA-decorated polymeric nanocarriers are a promising platform for targeted therapy in rheumatoid arthritis and possibly other autoimmune diseases characterized by HA receptor overexpression and local inflammation.
Comparison with Recent Literature and Novelty: MTX-loaded nanoparticles for targeted therapy have been studied in recent literature.9-11 We proceed with these endeavors by integrating HA functionalization with PLGA-PEG carriers to enhance receptor-specific targeting, structural stability, and anti-inflammatory activity, thereby demonstrating the innovativeness of the integrative design.
Limitations of the study: The current study did not consider in vivo biodistribution, long-term toxicity, or scaling, which should be addressed in future studies to confirm clinical applicability properly.
CONCLUSIONS
Hyaluronic acid-functionalized PLGA-PEG nanoparticles have been developed as an efficient nanocarrier system for targeted delivery of methotrexate in the treatment of rheumatoid arthritis. The engineered nanoparticles demonstrated optimal physicochemical characteristics, high encapsulation efficiency, and pH-responsive sustained drug release. Hyaluronic acid conjugation significantly increased anti-inflammatory activity and cellular uptake through CD44-mediated targeting of inflamed joints. Compared with free methotrexate, the HA–PLGA–PEG/MTX nanoparticles significantly reduced the severity of the collagen-induced arthritis model, inhibited the production of proinflammatory cytokines, and maintained joint architecture. These results show that site-specific drug delivery can be achieved while lowering systemic toxicity by combining nanomaterial engineering with immunopathological knowledge. This method provides a repeatable framework for creating polymeric nanocarriers to treat a range of inflammatory and autoimmune diseases.
Author Contributions: Marwa D. Jaaffer: Concept and design, writing, critical review of intellectual content, and final approval of the version to be published. Ammar M. Chaloop: Methodology, data curation, formal analysis, writing of the original draft, and interpretation of results. Sheimaa J. Hadi: Review and editing of the manuscript, interpretation of data, and final approval of the version to be published. All authors have read and approved the final version of the manuscript and agree to be accountable for all aspects of the work.
Funding: No specific grant from any public, private, or nonprofit funding agency was received for this study.
Institutional Review Board Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. This study did not involve human participants or animals; therefore, ethical committee approval was not required.
Informed Consent Statement: Not applicable.
Data Availability Statement: The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form. The authors declare no conflicts of interest.
AI-Assisted Tools Disclosure: The authors declare that no artificial intelligence tools were used to generate, analyze, or manipulate the scientific data or results presented in this study. If any AI-assisted tool was used for language improvement or figure preparation, such use was limited to editorial support under full human supervision, in accordance with the BioNatura Journal policy: https://bionaturajournal.com/artificial-intelligence--ai-.html
REFERENCES
1. Smolen JS, Aletaha D, McInnes IB. Rheumatoid arthritis. Lancet. 2016;388(10055):2023–2038. doi:10.1016/S0140-6736(16)30173-8
2. Firestein GS, McInnes IB. Immunopathogenesis of rheumatoid arthritis. Immunity. 2017;46(2):183–196. doi:10.1016/j.immuni.2017.02.006
3. Guo Q, Wang Y, Xu D, Nossent J, Pavlos NJ, Xu J. Rheumatoid arthritis: pathological mechanisms and modern pharmacologic therapies. Bone Res. 2018;6:15. doi:10.1038/s41413-018-0016-9
4. Cronstein BN, Aune TM. Methotrexate and its mechanisms of action in inflammatory arthritis. Nat Rev Rheumatol. 2020;16(3):145–154. doi:10.1038/s41584-019-0352-4
5. Singh JA, Saag KG, Bridges SL Jr, et al. 2019 update of the American College of Rheumatology guideline for the treatment of rheumatoid arthritis. Arthritis Care Res. 2019;71(6):684–699. doi:10.1002/acr.23780
6. Danhier F, Ansorena E, Silva JM, Coco R, Le Breton A, Préat V. PLGA-based nanoparticles: an overview of biomedical applications. J Control Release. 2012;161(2):505–522. doi:10.1016/j.jconrel.2012.01.043
7. Makadia HK, Siegel SJ. Poly(lactic-co-glycolic acid) (PLGA) is a biodegradable controlled drug delivery carrier. Polymers (Basel). 2011;3(3):1377–1397. doi:10.3390/polym3031377
8. Owens DE, Peppas NA. Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles. Int J Pharm. 2006;307(1):93–102. doi:10.1016/j.ijpharm.2005.10.010
9. Zhu S, Zhang T, Jiang C, et al. CD44-targeted hyaluronic acid-modified nanoparticles for rheumatoid arthritis therapy. Biomater Sci. 2022;10(4):934–947. doi:10.1039/d1bm01538k
10. Park JS, Choi J, Kim Y, et al. Methotrexate-loaded nanoparticles ameliorate experimental rheumatoid arthritis with reduced systemic toxicity. Int J Nanomedicine. 2022;17:809–824. doi:10.2147/IJN.S338421
11. Alexis F, Pridgen E, Molnar LK, Farokhzad OC. Factors affecting the clearance and biodistribution of polymeric nanoparticles. Mol Pharm. 2008;5(4):505–515. doi:10.1021/mp800051m
12. Benne N, van Duijn J, Kuiper J, Jiskoot W, Slütter B. Nanoparticles for inducing antigen-specific T-cell tolerance. Front Immunol. 2022;13:827815. doi:10.3389/fimmu.2022.827815
13. Hunter Z, McCarthy DP, Yap WT, et al. A biodegradable nanoparticle platform for antigen-specific immune tolerance. ACS Nano. 2014;8(3):2148–2160. doi:10.1021/nn406032q
14. Ghorpade VS, Yadav AV, Dias RJ. Development of methotrexate-loaded polymeric nanoparticles using solvent displacement. Pharm Nanotechnol. 2021;9(3):195–204. doi:10.2174/2211738509666210229115950
15. Li Z, Hu Y, Fang J, et al. pH-responsive release of methotrexate from PLGA nanoparticles for inflammation-triggered therapy. Acta Biomater. 2023;159:254–266. doi:10.1016/j.actbio.2023.01.025
16. Mosmann T. Rapid colorimetric assay for cellular growth and survival. J Immunol Methods. 1983;65(1–2):55–63. doi:10.1016/0022-1759(83)90303-4
17. Chen Y, Zhang Y, Yin Q, et al. Methotrexate-loaded chitosan nanoparticles suppress inflammatory mediators in activated macrophages. Carbohydr Polym. 2020;236:116017. doi:10.1016/j.carbpol.2020.116017
18. Zhang Q, Honko AN, Zhou J, et al. Nanoparticle modulation of macrophage polarization in autoimmune and inflammatory diseases. Nano Today. 2023;52:102021. doi:10.1016/j.nantod.2023.102021
19. Walvekar P, Ghadge SK, Patil S, et al. Hyaluronic acid-based nanocarriers for arthritis: current status and future perspectives. Int J Biol Macromol. 2023;235:123878. doi:10.1016/j.ijbiomac.2023.123878
20. Jaaffer MD, Abdulhussein EI, Abbas AW, Chaloop AM. Silicon dioxide nanoparticles enhance rice blast resistance in Oryza sativa. Agric Biotechnol J. 2026;18(1):339–362.
21. Brand DD, Latham KA, Rosloniec EF. Collagen-induced arthritis. Nat Protoc. 2007;2(5):1269–1275. doi:10.1038/nprot.2007.174
22. Beg S, Rahman M, Jain A, Saini S, Hasnain MS, Swain S. Nanoparticles for targeted drug delivery to rheumatoid arthritis: a comprehensive review. J Control Release. 2021;333:97–126. doi:10.1016/j.jconrel.2020.12.043
23. Li X, Hou Y, Zhang P, et al. pH-sensitive polymeric carriers for controlled drug delivery. J Mater Chem B. 2020;8:10072–10090. doi:10.1039/d0tb00992b
24. Niu J, Li Z, Yuan J, et al. pH-responsive nanocarriers for rheumatoid arthritis microenvironment. Adv Ther. 2022;5(2):2200014. doi:10.1002/adtp.202200014
Received: December 6, 2025 / Accepted: April 11, 2026 / Published (Online First): [Upload Date] / Issue Date: June 15, 2026 (Europe/Madrid)
Citation: Jaaffer MD, Hadi SJ, Chaloop AM. Hyaluronic Acid–Targeted PLGA Nanoparticles for Methotrexate Delivery in Rheumatoid Arthritis. BioNatura Journal: Ibero-American Journal of Biotechnology and Life Sciences. 2026;3(2):2. https://doi.org/10.70099/BJ/2026.03.02.2
Correspondence should be addressed to: marwa.dawood@kus.edu.iq
Peer Review Information: BioNatura Journal thanks the anonymous reviewers for their valuable contribution to the peer-review process. Regional peer-review coordination was conducted under the BioNatura Institutional Publishing Consortium (BIPC). Reviewer selection and assignment were supported via: https://www.reviewercredits.com/
Digital Preservation and Repository: This journal is managed through the Open Journal Systems (OJS) platform. To ensure long-term access, we use the PKP Preservation Network (PKP PN) to digitally preserve all published volumes in a decentralized, secure archive. Furthermore, our repository is integrated with LOCKSS and CLOCKSS, allowing international library networks to create permanent archives for long-term survival.
Publisher Information: Published by Clinical Biotec S.L. (Madrid, Spain) as the publisher of record under the BioNatura Institutional Publishing Consortium (BIPC). Places of publication: Madrid (Spain); Tegucigalpa (Honduras); Panama City (Panama). Online ISSN: 3020-7886.
