Tegoprubart and the CD40L Pathway: Promise and Remaining Questions in CNI-Free Kidney Transplantation
ABSTRACT
Calcineurin inhibitors (CNIs) remain the standard of care in kidney transplantation but are limited by chronic nephrotoxicity and high intra-patient pharmacokinetic variability (IPV), which drives late allograft loss. While costimulation blockade has emerged as a CNI-sparing alternative, the clinical adoption of belatacept is hindered by the logistics of intravenous administration, and recent receptor-targeting strategies, such as iscalimab (anti-CD40), have shown clinical limitations. This Perspective examines the re-emergence of CD40L as a viable therapeutic target through tegoprubart, an Fc-silent, subcutaneous anti-CD40L monoclonal antibody. We argue that targeting the ligand provides superior mechanistic pleiotropy compared with receptor blockade, more effectively suppressing T follicular helper cells and preventing endothelial prothrombotic activation. Furthermore, we highlight that tegoprubart’s subcutaneous delivery represents a critical pharmacokinetic advance, shifting maintenance immunosuppression from a behaviorally dependent daily regimen to a biological steady state. While early Phase 2 data (BESTOW trial) show promising renal preservation without de novo donor-specific antibody (DSA) formation, we caution that deep costimulatory blockade carries an inherent, long-term risk of opportunistic infection. The ultimate success of tegoprubart will depend not on 12-month efficacy, but on its ability to maintain host defense over a decade of use.
Keywords: Kidney transplantation; CD40L blockade; Calcineurin inhibitor-sparing; Tegoprubart; Costimulatory pathway; Intra-patient variability; Donor-specific antibodies.
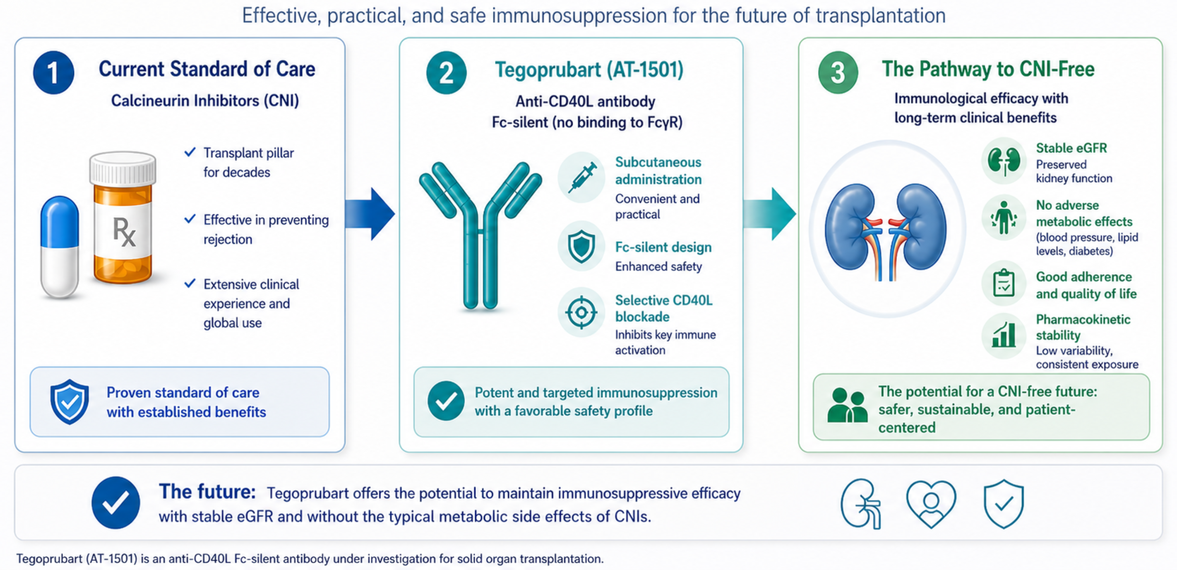
Graphical abstract. Shifting the renal transplantation paradigm: From CNI-based regimens to selective CD40L blockade. The schematic illustrates the transition from the current standard of care, centered on calcineurin inhibitors (CNIs) and their associated chronic nephrotoxicity, toward CNI-free strategies using tegoprubart. This anti-CD40L monoclonal antibody features a silent Fc domain to prevent thromboembolic events while maintaining robust costimulatory blockade. This approach aims to preserve long-term graft function by optimizing the eGFR profile and eliminating CNI-induced metabolic toxicities. Abbreviations: CNI, calcineurin inhibitor; CD40L, CD40 ligand; eGFR, estimated glomerular filtration rate; Fc, fragment crystallizable.
INTRODUCTION
The Shifting Paradigm of CNI-Sparing Immunosuppression
Calcineurin inhibitors (CNIs), particularly tacrolimus, remain the bedrock of maintenance immunosuppression, having revolutionized short-term graft survival. However, their chronic use exacts a severe toll: nephrotoxicity, neurotoxicity, and metabolic derangements, notably new-onset diabetes after transplantation (NODAT).¹,² While belatacept provided proof of concept for CNI-sparing via CD28/CTLA-4 blockade, its intravenous administration, risk of early acute rejection, and concerns about post-transplant lymphoproliferative disorder (PTLD) have limited its widespread adoption.³,⁴ Consequently, the field has looked downstream in the immune synapse to the CD40/CD40L axis.⁵
The era of CNI-free immunosuppression may have finally found its tipping point. Recent clinical setbacks with receptor-targeting strategies (e.g., iscalimab) have highlighted the biological limitations of blocking CD40.⁶ In contrast, tegoprubart (AT-1501)—a ligand-targeting, Fc-silent monoclonal antibody—has emerged not merely as another alternative, but as a potential paradigm shift.⁵,⁷ By rehabilitating the CD40L pathway through advanced protein engineering and offering subcutaneous administration, tegoprubart challenges the current clinical hierarchy.⁵,⁷,⁸,⁹ However, translating preclinical promise into long-term clinical reality requires navigating the complex legacy of costimulatory blockade.⁵,¹⁰
Box 1. The Evolution of Costimulatory Blockade in TransplantationGeneration 1 (Anti-CD40L + Fc active; e.g., Ruplizumab): Potent immunosuppression halted by catastrophic thromboembolic events due to Fc-receptor-mediated platelet activation.⁵,¹⁰Generation 2 (CTLA-4 Ig; e.g., Belatacept): First successful CNI-sparing agent, but burdened by intravenous logistics, higher early rejection rates, and PTLD risk.³,⁴Generation 3 (Anti-CD40L + Fc-silent; Tegoprubart): Decouples pathway inhibition from platelet activation. Adds subcutaneous delivery, aiming to combine the potency of Gen 1 with the safety and logistics required for modern outpatient care. ⁵,⁷,⁸,⁹
Mechanistic Rationale: Why the Ligand Outperforms the Receptor
The CD40/CD40L axis is a master regulator of adaptive immunity, orchestrating T-cell help, B-cell maturation, germinal center development, and donor-specific antibody (DSA) production.⁵ Initial clinical attempts to target CD40L in the early 2000s were derailed by thromboembolic complications, an effect later attributed to binding of Fc-gamma receptor (FcγRIIa) on platelets.⁵,¹⁰ Tegoprubart was engineered with an Fc-silent modification, effectively separating pathway inhibition from the safety profile that rendered first-generation molecules unviable.⁵,⁷
However, the renewed interest in CD40L is not just about safety engineering; it is about mechanistic superiority over receptor-directed approaches. The recent clinical shortcomings of iscalimab (anti-CD40) serve as a critical "cautionary tale" regarding the efficacy of receptor isolation.⁶ Blocking CD40 on the antigen-presenting cell (APC) leaves alternative CD40L interactions—such as those with CD11b on macrophages—intact.⁵ In contrast, CD40L blockade by tegoprubart provides a more proximal and pleiotropic interruption of the immune synapse. Preclinical models suggest that targeting the ligand more effectively suppresses T follicular helper (Tfh) cells and germinal center activity, thereby leading to a more profound reduction in de novo DSA formation (Figure 1).⁷
This mechanistic distinction is further validated outside standard allotransplantation. In the highly demanding realm of porcine-to-human xenotransplantation, CD40L blockade has become the immunological backbone for preventing hyperacute and acute humoral rejection, particularly by halting the prothrombotic activation of the graft endothelium—a primary barrier to recent xenotransplantation milestones.⁵,¹¹,¹² If CD40L inhibition is potent enough to bridge vast phylogenetic disparities and protect the endothelium, its application in standard kidney transplantation represents a logically robust, albeit clinically nascent, strategy.
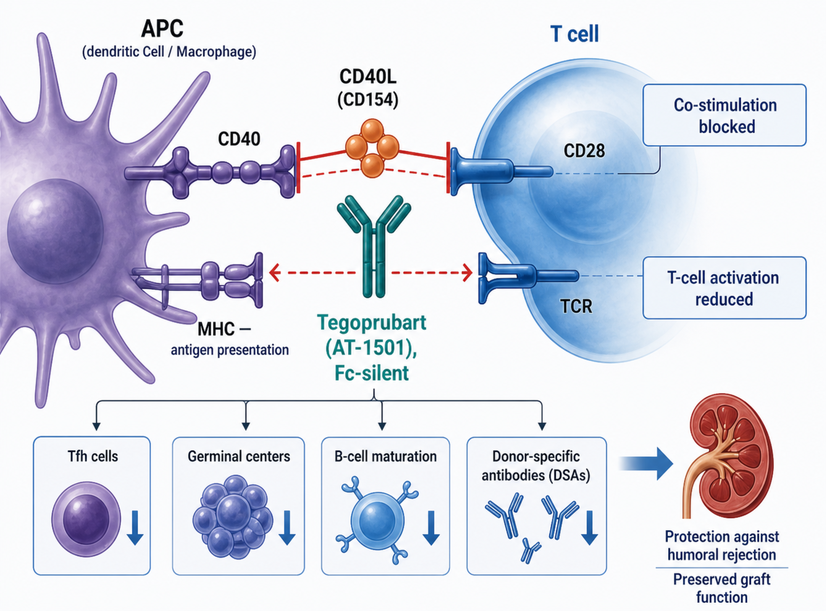
Figure 1. Mechanistic overview of tegoprubart-mediated CD40L blockade in kidney transplantation. Tegoprubart (AT-1501), an Fc-silent anti-CD40L monoclonal antibody, blocks CD40/CD40L-dependent co-stimulation, reducing T-cell activation, Tfh cell activity, germinal center responses, B-cell maturation, and donor-specific antibody formation. These effects may help protect against humoral rejection and preserve graft function.
The Subcutaneous Revolution: Eradicating Pharmacokinetic Valleys
Beyond mechanistic superiority, tegoprubart addresses a profound practical flaw in current maintenance therapy: pharmacokinetic (PK) variability. The reliance on daily oral tacrolimus creates notorious "troughs" in drug exposure and high intra-patient variability (IPV), which remains a leading cause of late allograft loss.² These subtherapeutic valleys are a primary driver of chronic antibody-mediated rejection (AMR) and de novo DSA formation, often exacerbated by patient non-adherence. Early pharmacokinetic data from Phase 1 trials and the ongoing BESTOW study (NCT05983770) demonstrate that tegoprubart’s subcutaneous formulation achieves sustained therapeutic concentrations.⁸,⁹ This shift to an SC-based maintenance regimen is not merely a convenience; it is a pharmacokinetic imperative to achieve therapeutic 'quiescence' in the immune synapse, bypassing the IPV inherent to oral CNIs. Conversely, belatacept, while avoiding PK valleys, demands monthly intravenous infusions, creating logistical bottlenecks and increasing costs for transplant centers.³,⁴ Tegoprubart bridges this remaining gap. By providing a biologic agent with a prolonged half-life in a rapid outpatient injection format, tegoprubart could stabilize immunosuppression in a way that daily pills and monthly infusions cannot. In a Perspective focused on the future of the field, this shift from behavioral dependence to biologically steady state may prove to be as clinically significant as the drug's mechanism of action itself.
Clinical Evidence: The BESTOW Program
Human data, while still early, are beginning to validate this preclinical promise. Interim 12-month data from the Phase 2 BESTOW trial demonstrate that tegoprubart-based regimens maintain stable estimated glomerular filtration rates (eGFR) without the early signals of tremor, NODAT, or de novo DSA formation that typically plague CNI-heavy regimens.⁹,¹³,¹⁴ While these results are derived from limited cohorts and require validation in larger Phase 3 trials, they represent the first successful translation of Fc-silent CD40L blockade into human kidney transplantation.
The Achilles' Heel: Immunologic Vigilance and the Infection Clock
However, maintaining scientific integrity requires acknowledging the inherent risks of deep costimulatory blockade. The transition from a 12-month safety signal to long-term clinical reality is fraught with peril. The CD40/CD40L pathway is critical for T-cell-dependent B-cell responses against viruses and malignancies.⁵
The field must heed the lessons from the 10-year follow-up of belatacept-based regimens, which demonstrated that while renal function is preserved, vigilance for cytomegalovirus (CMV), BK polyomavirus, and PTLD remains a lifelong necessity.¹⁵,¹⁶,¹⁷ Does the more profound pleiotropic inhibition of CD40L (compared to CTLA-4 Ig or anti-CD40) confer a higher risk of opportunistic infection? The early BESTOW cohorts have not shown overwhelming infectious signals, but as any transplant immunologist knows, immunosuppression safety is a marathon, not a sprint. If tegoprubart eliminates nephrotoxicity only to increase the incidence of severe viral reactivation in years 3 or 4, its clinical utility will be severely marginalized. Therefore, rigorous, long-term pharmacovigilance must be the cornerstone of the ongoing BESTOW extension trials (NCT06126380).¹⁸
CONCLUSIONS
As transplantation medicine pivots away from its historical reliance on calcineurin inhibitors, the re-emergence of CD40L as a viable therapeutic target stands as a triumph of protein engineering overcoming clinical history. Tegoprubart is not merely a toxicity-sparing agent; it represents a mechanistic pivot. By learning from the thromboembolic failures of first-generation anti-CD40L antibodies, the logistical limitations of belatacept, and the recent efficacy shortcomings of anti-CD40 receptor blockade, tegoprubart enters the clinical arena uniquely positioned. Its Fc-silent design and subcutaneous delivery offer an unprecedented opportunity to achieve therapeutic 'quiescence' at the immune synapse. However, the definitive validation of this molecule will not rest solely on preserving short-term renal functional milestones already achieved by previous biologics—but on its potential to redefine long-term graft survival without compromising host immune surveillance. The promise is unprecedented, but whether we are witnessing the end of the era of chronic nephrotoxicity depends on rigorous, decade-long observation.
Supplementary Materials: No supplementary materials are available for this article.
Author Contributions: Maria Cassola and Omar Junior Leal Niebla contributed to the conceptualization, literature review, critical analysis, manuscript drafting, and intellectual development of this Perspective article. Both authors contributed to the interpretation of the current evidence on CD40/CD40L blockade, tegoprubart, and CNI-free immunosuppressive strategies in kidney transplantation. Omar Junior Leal Niebla also assumed corresponding author responsibilities. Both authors have read and approved the final version of the manuscript and agree to be accountable for all aspects of the work.
Funding: This article received no external funding.
Institutional Review Board Statement: Not applicable. This article is a Perspective based on previously published literature, clinical trial reports, and publicly available scientific information and does not involve new studies with human participants, animals, or identifiable patient data.
Informed Consent Statement: Not applicable. This article does not include original patient data or identifiable clinical information.
Data Availability Statement: No new datasets were generated or analyzed during the preparation of this article. The discussion is based on previously published studies, conference reports, and publicly available clinical trial information cited in the reference list.
Acknowledgments: The authors acknowledge the broader transplant immunology and kidney transplantation research community whose work has contributed to the current understanding of CD40/CD40L pathway modulation, costimulatory blockade, and CNI-sparing immunosuppressive strategies.
AI-Assisted Tools Disclosure: The artificial intelligence tool GPAI (https://gpai.app/) was used solely to generate or refine the graphical representation included as Figure 1. No artificial intelligence system was used to generate, manipulate, or analyze experimental data, clinical data, statistical results, or scientific conclusions. The authors independently reviewed and verified the final content, interpretations, and conclusions, in compliance with the BioNatura Journal policy: https://bionaturajournal.com/artificial-intelligence--ai-.html
REFERENCES
1. Farouk SS, Rein JL. The many faces of calcineurin inhibitor toxicity—what the FK? Adv Chronic Kidney Dis. 2020;27(1):56-66. doi:10.1053/j.ackd.2019.08.006.
2. Schulz CA, Schütte-Nütgen K, Thölking G, Ehlert S, Möller K, Reuter S, et al. High intra-patient variability of tacrolimus exposure in the first year after kidney transplantation is associated with lower graft survival. Transplant Direct. 2020;6(11):e625. doi:10.1097/TXD.0000000000001058.
3. Vincenti F, Larsen CP, Alberu J, Bresnahan B, Garcia VD, Kothari J, et al. Three-year outcomes from BENEFIT, a randomized, active-controlled, parallel-group study in adult kidney transplant recipients. Am J Transplant. 2012;12(1):210-217. doi:10.1111/j.1600-6143.2011.03785.x.
4. Budde K, Prashar R, Haller H, Rial MC, Kamar N, Agarwal A, et al. Conversion from calcineurin inhibitor– to belatacept-based maintenance immunosuppression in renal transplant recipients: a randomized phase 3b trial. J Am Soc Nephrol. 2021;32(12):3252-3264. doi:10.1681/ASN.2021050628.
5. Singh AK, Goerlich CE, Zhang T, Lewis BG, D’Alessio FR. CD40-CD40L blockade: update on novel investigational therapeutics for transplantation. Transplantation. 2023;107(6):1239-1250. doi:10.1097/TP.0000000000004469.
6. Novartis. Novartis announces discontinuation of CIRRUS-1 study of CFZ533, iscalimab, in kidney transplant patients. 2021.
7. Anwar IJ, Berman DM, DeLaura I, et al. The anti-CD40L monoclonal antibody AT-1501 promotes islet and kidney allograft survival and function in nonhuman primates. Sci Transl Med. 2023;15(711):eadf6376. doi:10.1126/scitranslmed.adf6376.
8. ClinicalTrials.gov. Phase 1b study of AT-1501 in kidney transplant recipients. NCT05027906.
9. ClinicalTrials.gov. BESTOW: safety and efficacy of tegoprubart in kidney transplantation. NCT05983770.
10. Boumpas DT, Furie R, Manzi S, Illei GG, Wallace DJ, Balow JE, et al. A short course of BG9588, anti-CD40 ligand antibody, improves serologic activity and decreases hematuria in patients with proliferative lupus glomerulonephritis. Arthritis Rheum. 2003;48(3):719-727. doi:10.1002/art.10856.
11. Moazami N, Stern JM, Khalil K, Kim JI, Narula N, Mangiola M, et al. Pig-to-human heart xenotransplantation in two recently deceased human recipients. Nat Med. 2023;29(8):1989-1997. doi:10.1038/s41591-023-02471-9.
12. Montgomery RA, Stern JM, Lonze BE, Tatapudi VS, Mangiola M, Wu M, et al. Results of two cases of pig-to-human kidney xenotransplantation. N Engl J Med. 2022;386(20):1889-1898. doi:10.1056/NEJMoa2120238.
13. Katz E, Adams AB, et al. Safety and preliminary efficacy of tegoprubart in kidney transplantation: results from the BESTOW Phase 1b/2 study. Presented at: American Transplant Congress; 2024.
14. Adams AB, et al. Efficacy and safety of tegoprubart for the prevention of rejection in kidney transplantation: results from the Phase 2 BESTOW trial. J Am Soc Nephrol. 2025;36(10S):TH-OR089.
15. Vincenti F, Charpentier B, Vanrenterghem Y, Rostaing L, Bresnahan B, Darji P, et al. Ten-year outcomes in a randomized phase II study of kidney transplant recipients administered belatacept 4-weekly or 8-weekly. Am J Transplant. 2017;17(12):3219-3227. doi:10.1111/ajt.14416.
16. Terrec F, Jouve T, Malvezzi P, Noble J, Janbon B, Naciri-Bennani H, et al. Belatacept use after kidney transplantation and its effects on risk of infection and COVID-19 vaccine response. J Clin Med. 2021;10(21):5159. doi:10.3390/jcm10215159.
17. Bertrand D, Chavarot N, Gatault P, Garrouste C, Bouvier N, Grall-Jezequel A, et al. Opportunistic infections and efficacy following conversion to belatacept-based therapy after kidney transplantation: a French multicenter cohort. J Clin Med. 2020;9(11):3479. doi:10.3390/jcm9113479.
18. ClinicalTrials.gov. BESTOW-EXTENSION: long-term safety and efficacy of tegoprubart in kidney transplant recipients. NCT06126380.
Received: March 12, 2026 / Accepted: May 11, 2026 / Published (Online First): May 11, 2026 / Issue Date: June 15, 2026 (Europe/Madrid)
Citation: Cassola M, Leal Niebla OJ. Tegoprubart and the CD40L Pathway: Promise and Remaining Questions in CNI-Free Kidney Transplantation. BioNatura Journal: Ibero-American Journal of Biotechnology and Life Sciences. 2026;3(2):10. https://doi.org/10.70099/BJ/2026.03.02.10
Correspondence should be addressed to: marycaryrn@gmail.com
Peer Review Information: BioNatura Journal thanks the anonymous reviewers for their valuable contribution to the peer-review process. Regional peer-review coordination was conducted under the BioNatura Institutional Publishing Consortium (BIPC). Reviewer selection and assignment were supported via: https://www.reviewercredits.com/
Digital Preservation and Repository: This journal is managed through the Open Journal Systems (OJS) platform. To ensure long-term access, we use the PKP Preservation Network (PKP PN) to digitally preserve all published volumes in a decentralized, secure archive. Furthermore, our repository is integrated with LOCKSS and CLOCKSS, allowing international library networks to create permanent archives for long-term survival.
Publisher Information: Published by Clinical Biotec S.L. (Madrid, Spain) as the publisher of record under the BioNatura Institutional Publishing Consortium (BIPC). Places of publication: Madrid (Spain); Tegucigalpa (Honduras); Panama City (Panama). Online ISSN: 3020-7886.
