Phytochemical evaluation and biological activity of three plant extracts from Cumana, Venezuela
1 Yachay Tech Medicinal Chemistry Research Group (MedChem-YT), School of Chemical Sciences and Engineering, Yachay University for Experimental Technology and Research (Yachay Tech), Urcuquí 100119, Ecuador.
E-mails: vmorera@yachaytech.edu.ec (VMC);hrodriguez@yachaytech.edu.ec (HRC).
E-mails: vmorera@yachaytech.edu.ec (VMC);hrodriguez@yachaytech.edu.ec (HRC).
2 Universidad de Oriente, Núcleo de Sucre, Escuela de Enfermería, Departamento de Bioanálisis, Cumaná 6101, Venezuela.
E-mails: yjrivas17@gmail.com (YRC); hherrera.mata@gmail.com (HHM).
E-mails: yjrivas17@gmail.com (YRC); hherrera.mata@gmail.com (HHM).
Correspondence: Shailili Moreno Morales; smoreno@yachaytech.edu.ec
ABSTRACT
Kalanchoe pinnata, Lantana camara L., and Melia azedarach L. are common plants in Cumana, Venezuela, and are traditionally used by locals to treat infections. This research aimed to evaluate the phytochemical profile and antibacterial and cytotoxic activities of the ethanolic leaf extracts from these plants. Specimens were collected and identified. Using qualitative colorimetric and precipitation tests, the profile of secondary metabolites in the ethanolic extract of the leaves of each plant was determined. These metabolites were confirmed by FT-IR and UV-visible spectroscopies. Antimicrobial and cytotoxic activities were evaluated using the disk diffusion method and the Artemia salina model, respectively. All three extracts contain coumarins and polyphenols. K. pinnata and Lantana camara L. contain unsaturated sterols, but alkaloids and saponins were only detected in L. camara. None of the extracts exhibited antibacterial activity at the concentrations tested. However, according to the Clarkson Toxicity Index, all extracts showed medium toxic activity (LC50: 100–500 µg mL-1) against A. salina nauplii, with LC50 values of 245.20, 261.27, and 151.40 µg∙mL⁻¹ for K. pinnata, L. camara, and M. azedarach, respectively. These results demonstrate that the plants studied are a potential source of polar compounds with biologically active properties.
Keywords: Kalanchoe pinnata, Lantana camara, Melia azedarach, metabolites, Venezuelan plants, biological activity.
INTRODUCTION
Plants have the capacity to manufacture a great variety of organic products of low molecular weight from simple inorganic precursors, among them phenolic compounds, terpenes, and alkaloids. This capacity for synthesis has been essential for their reproduction, colonization of diverse environments, establishment of symbiotic relationships, and, especially, their survival. Commonly, these compounds are referred to as secondary or specialized metabolites, phytochemicals, or natural products.1
Secondary metabolites have been of great interest due to their multiple biological activities: specialized metabolites are essential for the treatment of various infections and diseases. Today, they are the basis of a significant number of drugs.2,3,4 In addition, secondary metabolites also have industrially remarkable uses, as many of them are valuable for the production of insecticides, cosmetics, food products, and nutraceuticals, among others.5,6
The gap in the study of secondary metabolites in new plants lies in the need to discover, characterize, and understand the functions of thousands of compounds that remain unknown for various applications. Although many are known, thousands of secondary metabolites with ecological functions and potential applications remain underexplored, particularly in newly described or understudied species. Furthermore, there remains a need to develop efficient, sustainable methods for producing these compounds at an industrial scale, as traditional cultivation can be limited or inefficient. Likewise, research on how environmental factors and biotic stress activate or inhibit the production of these metabolites in unstudied plants, and how to manipulate these responses, is insufficient. A better understanding of the complex plant-microorganism-herbivore interactions at the molecular level, which is key to designing more ecological pest control strategies, is also important. For all these reasons, the description of the secondary metabolites present in plants is vital, and this work contributes to this for the species mentioned.
In this sense, the Sucre state is located in the mountain range of the eastern coast of Venezuela, with an area of 11,800 km2. It is bordered on the north by the Caribbean Sea, on the south by the states of Monagas and Anzoátegui, on the east by the Gulf of Paria, open to the Atlantic Ocean, and on the west by the Gulf of Cariaco. Its geographic location contributes to a highly varied relief, predominantly mountainous, with flora characteristic of the state of Sucre.7,8
Within the flora of Sucre State, in eastern Venezuela, there are abundant Kalanchoe pinnata, Lantana camara, and Melia azedarach, which, belonging to different families, are used by local inhabitants for the same purpose. They use their ethnobotanical knowledge to cure ailments associated with several bacterial diseases and cancer. Studies on K. pinnata demonstrate antifungal 9, antibacterial, and antioxidant10 activity in its leaves, and it selectively inhibits cell proliferation in colon cancer11. Similarly, there is evidence of the bioactivity of L. camara, highlighting its insecticidal12 and medicinal13 actions in the treatment of wounds and ulcers. M. azedarach is also considered a natural insecticide14 and has broad-spectrum antibacterial properties against pathogenic bacteria15.
The objective of this study was to determine whether metabolites present in ethanolic extracts of leaves from three plants (Kalanchoe pinnata, Lantana camara, and Melia azedarach), collected in Sucre, Venezuela, exhibit antibacterial activity against Gram-positive and Gram-negative strains and cytotoxicity toward Artemia salina. The results obtained provide useful information on the potential therapeutic properties of these species.
MATERIAL AND METHODS
Plant material and the obtention of extracts
Specimens of Lantana camara L., Melia azedarach L., and Kalanchoe pinnata were sampled in the city of Cumana (Longitude: W64°10'57.22", Latitude: N10°27'14. 29"), Sucre state, Venezuela. The plants were identified at the Isidro Ramon Bermudez Romero herbarium (HIRBR) of the Biology Department of the Universidad de Oriente, Núcleo de Sucre (UDO-NS). Then, plant samples were transferred to the Laboratory of Natural Products and Lipids (EC-310) of the Chemistry Department at UDO-NS. The leaves of each species, free of any visible disease, were washed under a constant flow of common water and then distilled water, cut into small pieces, and dried on blotting paper, at room temperature, in the shade.
The dried plants were weighed and pulverized separately in an electric mill. The extracts were obtained by solid-liquid extraction (maceration) in ethanol 95%, using a solvent-to-solid ratio of 10 mL/1 g, until the solvent was exhausted of color. The solution obtained from each plant extract was filtered and evaporated to dryness under reduced pressure in a Heidolph rotary evaporator at 50 °C. The recovered solvent was used again in the extraction process. Finally, the extracts were transferred to vials of known weight, placed in a desiccator at room temperature until use. For reference of each extract in this work, the following identifiers are used: KE: leaves of K. pinnata, LE: leaves of L. camara, ME: leaves of M. azedarach.
Phytochemical analysis of the crude extracts
To detect secondary metabolites in the plant extracts, specific chemical tests were performed according to the phytochemical analysis methodology.16,17
Alkaloids
A portion of the extract (2.5mg) was dissolved in 10% HCl, then shaken with chloroform, and the phases were separated. The aqueous phase was alkalinized with a dilute NaOH solution and partitioned again with chloroform. The three phases were analyzed separately with Dragendorff's reagent for weakly basic alkaloids, basic alkaloids, and quaternary ammonium salts, respectively. A red-orange coloration was considered a positive result for the possible presence of alkaloids.
Anthraquinones
The extract (1mg) was treated with 0.5 mol L-1 KOH, then acidified with acetic acid and subsequently extracted with benzene. The organic layer was separated and alkalinized with NH4OH; a red coloration indicated the possible presence of anthraquinones.
Cumarins
In a test tube, 1 mg of the extract was dissolved in ethanol, covered with filter paper soaked in a dilute NaOH solution, and then placed in a water bath at 100°C for several minutes. Subsequently, when the filter paper was removed and observed under UV light, a yellow fluorescence indicated the possible presence of coumarins.
Unsaturated sterols and pentacyclic triterpenes
The extract (1 mg) was dissolved in chloroform, and a few drops of Liebermann-Burchard reagent were added. A blue to green coloration in the solution indicated the presence of sterols, while a red to violet coloration is considered positive for triterpenes, and yellow to orange colorations indicate the possible presence of sterols and triterpenes.
Flavonoids
1 g of the crude extract was defatted with petroleum ether, filtered and the residue was separated for analysis, using the following method: in a test tube the residue was mixed with 2 mL of concentrated HCl and magnesium shavings, considering the result positive for flavonoids, when a red coloration was produced when the reaction was left to stand for 10-20 minutes.
Cyanogenic and cardiotonic glycosides
The extract was dissolved in chloroform (1 mg mL-1) in a test tube, and the mixture was heated in a bath at 50-70°C. The vapors were brought into contact with filter paper impregnated with a 1% solution of picric acid in 10% sodium carbonate. The cyanogenic compounds appear as a red stain on the paper.
Cardiotonic glycosides were detected by reaction with a freshly prepared 1:1 mixture of 3,5-dinitrobenzoic acid (2%) and KOH (0.5 mol · L-1); if present, a blue or violet coloration occurred.
Methylene ketones
The crude extract (1mg) was dissolved in distilled water, and then a few drops of Baljet's reagent (1:1 mixture of 1 g picric acid in 25 mL ethanol and 2 g NaOH in 25 mL water) were added. A red coloration indicated the possible presence of the metabolite.
Tannins and polyphenols
A portion of the extract was dissolved in distilled water (1mg mL-1), and phenolic compounds were detected by the brown color they produced in the presence of a 1% ferric chloride solution, resulting in a dark brown or black color, which was considered a positive result. Tannins were identified by treating the extract with a 1% gelatin solution in 1% NaCl; the formation of a white precipitate indicated the possible presence of the compound.
Saponins
The presence of saponins was indicated by the formation of a persistent foam for 10 minutes, when a small portion of the extract (0.5mg mL-1) was shaken vigorously with distilled water. Additionally, saponins were detected using the red blood cell hemolysis test, in which a concentrated extract solution was prepared and then mixed with 3.5 mL of human blood in a test tube. The tubes were centrifuged at 1500 rpm for 5 min.
Spectroscopic analysis
The presence of secondary metabolites in the ethanolic extracts was confirmed by conventional spectroscopic techniques, such as Fourier transform infrared spectroscopy (FT-IR) and ultraviolet-visible spectroscopy (UV-vis).
For the analysis of the extracts by FT-IR, a white tablet with KBr was used, and then the dried samples were mixed with KBr powder and crushed in an agate mortar to homogenize the mixture, the tablets were compacted in a press to obtain a translucent disc, which was placed in a Perkin-Elmer Frontier instrument, where the spectra were obtained in a wavenumber range of 400 to 4000cm-1, accumulating 24 scans, with a resolution of 2cm-1. The assignment criteria were based on the analysis of molecular vibrations (peaks) in each spectrum to assign functional groups.
The extracts (0.1mg mL-1 in ethanol) were analyzed by UV-Vis spectroscopy using a JENWAY model 6405 spectrophotometer to determine the chromophores present, according to the criteria for qualitative analysis of absorption peaks. The solvent was used as a blank, and the spectral range was 200-450 nm.
Evaluation of the biological activity of crude extracts
Antibacterial activity
The antibacterial activity was determined using four certified strains, two of them Gram-positive: Staphylococcus aureus ATCC 25923 and Enterococcus faecalis ATCC 29212, and two Gram-negative: Pseudomonas aeruginosa ATCC 27853 and Escherichia coli ATCC 25922. All strains used in this study were donated by the Bacteriology Laboratory of the Antonio Patricio Alcalá University Hospital (HUAPA). To perform this bioassay, the agar diffusion technique was used. Sterile filter paper 5 mm diameter discs (Whatman No. 3) were impregnated with 10 µL of a 30, 40, and 60 mg∙mL-1 solution of the extract. Commercial Vancomycin for Gram-positive strains and commercial Ofloxacin for Gram-negative strains were used as positive controls, and ethanol was used as the negative control. The discs were placed in a Petri dish containing Müller-Hinton agar, previously inoculated with a standardized bacterial suspension (McFarland 0.5). Subsequently, the plates were pre-incubated at 5 °C for 24 hours, followed by incubation at 37 °C for 24 hours. Each assay was performed in triplicate, and antibacterial activity was determined by measuring the diameter (mm) of the bacterial growth inhibition halo around the impregnated disc.18
Cytotoxic activity against Artemia salina
To perform this assay, A. salina cysts were initially placed in bifiltered seawater, contained in a 1 L Erlemeyer flask, with continuous aeration and artificial light for 24 hours, to obtain the crustacean nauplii. On the other hand, a stock solution of 10. 000 µg∙mL-1 of the extract in 0.5 mL of dimethyl sulfoxide (DMSO) and 4.5 mL of bifiltered seawater, from this, solutions with concentrations of 1000; 100; 10; 1; successive dilutions prepared 0.1 and 0.01 µg∙mL-1; the process was repeated by preparing a stock solution without the extract, which served as a negative control, and solutions of Vimblastine sulfate were used as a positive control; each dilution was performed in triplicate. Subsequently, 10 nauplii of A. salina hatched 24 hours in advance were added to each solution. The quantification of nauplii mortality was carried out after 24 hours, and the final analysis of cytotoxic activity was performed using the Binomial statistical methods designed to determine the mean lethal concentration (LC50).19
RESULTS
Ethanol is a solvent commonly used in the extraction of plant materials for medicinal purposes, and it is effective at extracting various bioactive compounds, many of which exhibit antimicrobial, antioxidant, and anticancer activity, as demonstrated by reports in the scientific literature. Even previous research on the plants selected for this study used ethanol. 9,11,20.
Extraction yield
After plant collection, identification, and processing, ethanolic leaf extracts were obtained. The extraction yields were 27.61%, 10.45%, and 17.16% for KE, LE, and ME, respectively (Table 1).
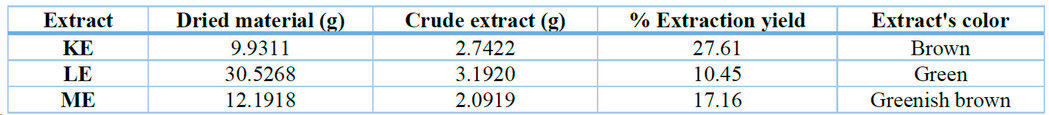
Table 1. Ethanolic extraction yield from the leaves of K. pinnata (KE), L. camara (LE), and M. azedarach (ME).
Phytochemical analysis of the extracts
Coumarins, polyphenols, and unsaturated sterols were detected in the ethanolic extract of K. pinnata leaves (KE extract). The ethanolic extract of L. camara (LE extract) showed variability in its metabolite profile in phytochemical studies. Alkaloids, coumarins, polyphenols, saponins, and unsaturated sterols were detected in this extract. Finally, coumarins and polyphenols were identified in the leaf extract of M. azedarach (ME extract) (Table 2).

(+): metabolite detected; (-): metabolite not detected
Table 2. Phytochemical analysis performed on ethanolic extracts of the leaves of Lantana camara L., Melia azedarach L. and Kalanchoe pinnata.
Spectroscopic analysis
UV-vis spectra showed several adsorption maxima in the region between 200 nm and 400 nm. For the KE extract, the most relevant peaks were observed at 230 nm, 265 nm, and 350 nm. In the ME extract, peaks were observed at 225 nm, 270 nm, and 360 nm. Finally, in the ME extract, absorption maxima were identified at 230 nm, 290 nm, and 335 nm (Figure 1).
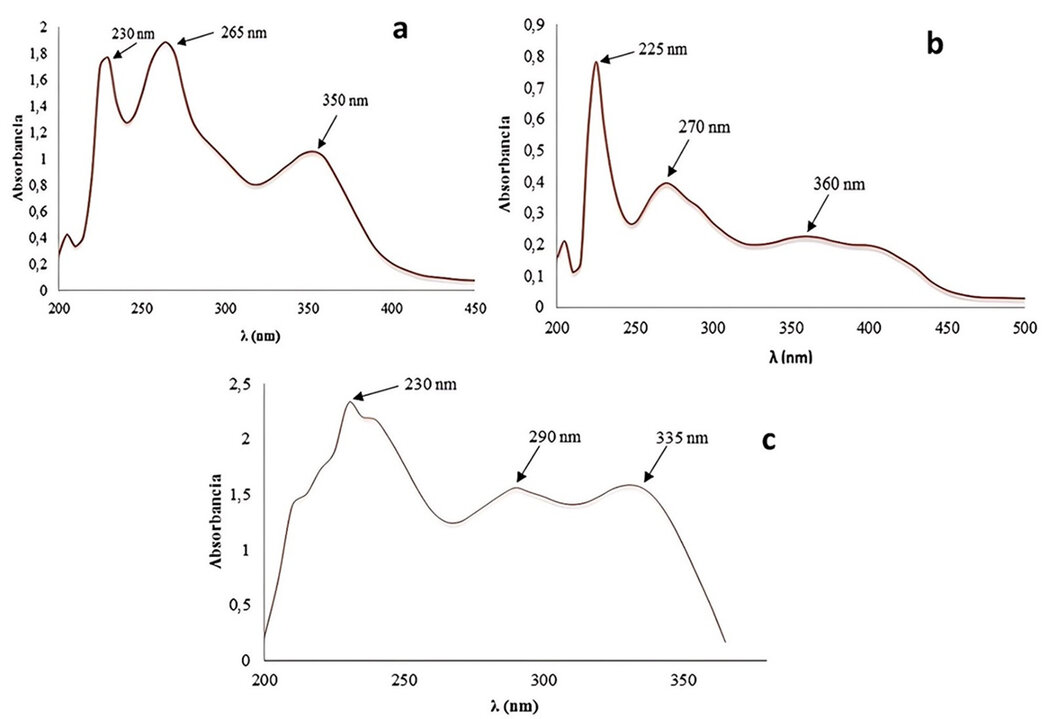
Figure 1. UV-vis spectra. (a) Ethanolic extract KE from K. pinnata, (b) Ethanolic extract ME from M. azedarach, and (c) Ethanolic extract from L. camara.
Fourier Transform Infrared Spectroscopy (FT-IR) was used to identify the functional groups of the active components by detecting specific molecular vibrations via their infrared absorption peaks. The obtained FT-IR spectra are shown in Figure 2.

Figure 2. Infrared spectra of the ethanolic extract of K. pinnata (a), M. azedarach (b), and L. camara (c).
Comparing the signals in each spectrum with the average theoretical values (FT-IR21, 22; UV-vis23) enabled the establishment of specific relationships among the functional groups of the metabolites identified in the species studied, thereby confirming their presence in the extracts (Table 3).

* 360 nm reportado como banda débil/indefinida (nota al pie).
Table 3. Functional-group identification by metabolite class in ethanolic leaf extracts of Kalanchoe pinnata, Melia azedarach, and Lantana camara based on theoretical FT-IR/UV-Vis signals and experimental bands (FT-IR, cm⁻¹; UV-Vis, λmax in nm). ND: not detected.
Evaluation of the biological activity of crude extracts
The results of the antibacterial activity at different concentrations (Table 4) and the toxic activity (Table 5) against A. salina of the extracts from the three plant species tested are presented below.

NI: did not inhibit, VAN: Vancomycin, an antibiotic used as positive control for Gram-positive bacteria, and OFL: Ofloxacin, an antibiotic used as positive control for Gram-negative bacteria. 1: 30 mg∙mL-1, 2: 40 mg∙mL-1, 3: 60 mg∙mL-1
Table 4. Antibacterial activity of ethanolic extracts of K. pinnata, L. camara, and M. azedarach leaves at different concentrations.

Table 5. Mortality percentage, median lethal concentration, and Clarkson's toxicity of plant extracts against the crustacean Artemia salina.
DISCUSSION
Plant material and its extracts
Of the three ethanolic extracts obtained, K. pinnata has the highest mass yield (Table 1), indicating that this plant synthesizes a greater number of polar, alcohol-soluble compounds than the other two species under study. The color that characterizes each extract suggests that the structure of some of its constituents has chromophore groups, capable of absorbing energy in the UV-Visible region. 24
Phytochemical analysis of the extracts
Phytochemical profiling is the analysis, using chemical reactions that target specific functional groups, that enables the identification of secondary metabolites present in a plant. The study of the phytochemical profile enables evaluation of a plant's therapeutic potential, as many phytochemicals possess medicinal properties and serve as targets for the development of new drugs or supplements.
Likewise, knowledge of a plant's phytochemical profile enables the definition of quality standards for herbal drugs. In biological research, understanding a plant's phytochemical composition helps elucidate its responses to biotic and abiotic stressors and its adaptation to the environment. In addition, knowledge of a plant's phytochemical profile enables the selection of extraction protocols for the isolation of pure secondary metabolites and their subsequent structural characterization.
To initiate an active synthesis of secondary metabolites, plants must be exposed to adverse conditions such as consumption by herbivores (arthropods and vertebrates), attack by microorganisms (viruses, bacteria and fungi), competition for soil space and light and nutrients between different plant species, and exposure to sunlight or other abiotic stresses, which explains why a plant prefers to synthesize or not certain metabolites as a function.25 Alkaloids, polyphenols, coumarins, saponins, and unsaturated sterols were detected in the ethanolic extracts (Table 2).
Alkaloids are organic bases with a large structural variety.26 Previous research reports the presence of alkaloids in extracts of M. azedarach using Dragendorff's reagent, for such metabolites in fruits, flowers, stem, and bark that confer insecticidal properties to M. azedarach known as paraisin and azadirachtin.20 However, the ethanolic extract of this species did not show the presence of alkaloids.
But a positive result was obtained for the extract of L. camara, coinciding with recent reports of tests performed on this plant in Brazil, which showed positivity for alkaloids.27
On the other hand, phenols were detected in the three extracts tested (Table 2); in this sense, it is known that plants synthesize a great variety of secondary products containing a phenol group; these substances are called phenolic compounds or polyphenols, and all of them are derived from phenol,28 supporting the results obtained in the tests performed on the extracts under study.
Regarding coumarins, all three extracts tested positive. These compounds, α-pyrones, are widely distributed in the plant kingdom and occur in seeds, leaves, and roots. This result coincides with those reported by Ramos and collaborators, who qualitatively detected coumarin in an extract of M. azedarach,14 and by Navarrete, who reported this metabolite from the extraction of a L. camara specimen.29 The presence of coumarins in these plants, and their possible association with phenols such as hydrous cimarins, may be related to the biological activity of the extracts, since reports of coumarins as antioxidants profile them as efficient substances against chronic degenerative diseases, heart disease, and other ailments such as diabetes and even tumor diseases.30
Sterols also gave positive results in two of the extracts tested. These metabolites are widely distributed in the animal and plant kingdoms; they occur in free form (also called steroid aglycones), as esters, or as glycosides. A large number and diversity of substances with the steroid nucleus are found in nature. Most natural sterols or unsaturated sterols have a side chain of 8 to 10 carbon atoms and a double bond at C-5 in the case of the cholesterol molecule.31
Moreno et al. reported the presence of sterols in the extracts of K. pinnata, and likewise, Amaya, in his study of M. azedarach, determined the presence of sterols in the plant, 32,33 results that coincide with those reported in this research, in which their possible presence was detected in the extract obtained by maceration of the leaves of K. pinnata.
Spectroscopic analysis
The intense coloration that characterized each extract suggests that the structure of some of its constituent metabolites possesses chromophore groups capable of absorbing energy in the UV-visible region.
For this reason, this technique was used to identify secondary metabolites containing π-bonds, σ-bonds, lone pairs of electrons, aromatic rings, and other chromophores across the electromagnetic spectrum from 200 to 750 nm. In the UV spectrum of the L. camara extract obtained by maceration in ethanol (Figure 1c), maximum absorption bands corresponding to electronic transitions of the chromophore groups present in the extract are observed. The signal at 230 nm corresponds to electronic transitions, indicating the presence of polyphenols and confirming the preliminary chemical analysis results. On the other hand, the signal at 290 nm is attributed to electronic transitions of the C=O chromophore in the coumarins and is corroborated by the signal at 335 nm, which is a radical band characteristic of atoms with unshared electron pairs.34
Similarly, in the UV spectra for K. pinnata and M. azedarach (Figure 1a and 1b, respectively), characteristic signals belonging to the metabolites identified in their extracts were identified, highlighting the fact that in K. pinnata, the band defined at 350 nm corresponds to the positive result of alkaloid in its extract, since the structures containing the quinoline skeleton present an excellent chromophore, in the region between 300 to 400 nm.35 Whereas, in M. azedarach, which has the extract with the least variability of metabolites, this band is practically imperceptible, and only the one at 225 (polyphenols) and 270 nm (C=O of coumarins) is appreciated.
Similarly, in the UV spectra for K. pinnata and M. azedarach (Figure 1a and 1b, respectively), characteristic signals belonging to the metabolites identified in their extracts were identified, highlighting the fact that in K. pinnata, the band defined at 350 nm corresponds to the positive result of alkaloid in its extract, since the structures containing the quinoline skeleton present an excellent chromophore, in the region between 300 to 400 nm.35 Whereas, in M. azedarach, which has the extract with the least variability of metabolites, this band is practically imperceptible, and only the one at 225 (polyphenols) and 270 nm (C=O of coumarins) is appreciated.
FT-IR spectroscopy is an analytical technique used to identify functional groups in secondary metabolites. This technique is widely used in the investigation of plant chemical composition. FT-IR spectroscopy has experienced rapid development thanks to its low noise level, high analytical speed, high repeatability, easy operation, and low cost.22
Likewise, bands attributable to phenolic compounds were observed in the infrared spectrum of the LE extract (Figure 2c). The band at approximately 3395 cm-1 corresponds to the stretching of the O-H bond of alcohols, phenols, or carbohydrates associated with saponins, although this zone also corresponds to the N-H tension of alkaloidal compounds. In contrast, the band at 2929 cm-1 is a characteristic signal of C-H bond vibration, corresponding to alkane-type systems or saturated hydrocarbons, which are normally present in sterols as side chains of 8 to 10 carbon atoms. 21 A broad band with small peaks at 1545 cm-1 (C-H and C-C stretching) and 1600 cm-1 (C=N, C=O and C=C vibration zone) is also seen. The signal at 1164 cm-1 indicates the presence of C-O bonds of sugar units (carbohydrates) mentioned in association with saponins detected in the preliminary chemical analysis. On the other hand, a band at 1073 cm-1 is attributed to the C-O bond stretching vibration of primary alcohols and phenols respectively, corresponding also to C-N single bond vibration; on the other hand, the band at 1719 cm-1 concerns the C=O bond indicating the presence of coumarins and the band emitted at 817 cm-1 can be attributed to the bending of the C-H bond corresponding to a poly substituted aromatic ring.
In the infrared spectrum of the KE extract (Figure 2a), the signals corresponding to the main functional groups found are observed, such as polyphenols evidenced by a broad and intense band ~3403 cm-1 that is related to the O-H stretching of alcohols and phenols and, as in L. camara, the band at 2928 cm-1 is characteristic of C-H bond vibration corresponding to alkane or saturated hydrocarbon type systems, normally present in sterols, as a side chain of 8 to 10 carbon atoms.24 The signal at 1402 cm-1 is attributed to C-C bond bending. Similarly, the band at 1065 cm-1 corresponds to C-O stretching of phenols or sterols since they possess at least one -OH group. On the other hand, the signals found at 1702 cm-1 and 1560 cm-1 correspond to the stretching and vibration of the C=O double bond of carbonyl groups, suggesting the presence of coumarins; while the signal observed at 776 cm-1 may indicate C-H bending out of the plane corresponding to a p-disubstituted aromatic ring.
Finally, in the phytochemical analysis of the ME leaf extract, only the possible presence of coumarins and polyphenols was detected, finding in the IR spectrum (Figure 2c) a broad and intense band between 3500 and 3100 cm-1 where the O-H bond corresponding to alcohols and phenols is reflected; taking into account the signal manifested at 1384 cm-1 that alludes to the C=O bond, indicative of the presence of coumarins in the extract, and it can also be observed that the band at 1623 cm-1 corresponding to the stretching of the C=C bond indicates the presence of an instauration.
Evaluation of the biological activity of crude extracts
Antibacterial activity
Antibacterial activity is one of the most significant bioassays in the pharmacological study of a substance. Due to the continuous growth of bacterial resistance caused by the indiscriminate use of common antibiotics, bacterial resistance is a natural biological phenomenon, where a drug inhibits bacteria, but is not destroyed by it, this has resulted in a problem for the treatment of infectious diseases;36 therefore, research is currently being carried out in the search for new compounds with bactericidal properties.
Gram-negative bacteria are generally more resistant to antiseptics and disinfectants than Gram-positive bacteria. The outer membrane of Gram-negative bacteria acts as a barrier that limits the entry of several types of chemically unrelated antibacterial agents. Hydrophilic molecules of low molar mass pass easily through porins, whereas hydrophobic molecules diffuse through the membrane bilayer. Gram-positive bacteria have only a cell membrane with a broad outer peptidoglycan layer. These characteristics of each bacterial structure largely define their permeability to antibiotic agents and the transport of molecules across barriers, being important factors to consider in the case of Gram-negative bacteria.37
According to the antibiogram results, the extracts showed no antibacterial activity against the strains tested. This lack of activity could be due to the medium's low diffusion capacity. It should also be noted that these are crude extracts containing multiple components, probably at low concentrations, insufficient to produce a bactericidal effect.37 A study of the hydroalcoholic extract of K. pinnata showed significant biological activity;38 however, in this investigation, extracts of the same plant did not exhibit any activity against the bacteria tested.
Lethal activity against Artemia salina
The brine shrimp lethality assay combines practicality, cost-effectiveness, and simplicity into a single method for assessing the toxicity of plant extracts. These biological effects of natural compounds often correlate strongly with cytotoxic and antitumor activities. 39,40 The desired biological reaction typically results from the combination of bioactive plant components rather than from a single component. Therefore, a medicinal plant extract often exhibits cytotoxic effects against brine shrimp larvae with a concentration-dependent activity profile 41, and the compounds responsible for the extract's toxicity may be antitumor agents.42
The Artemia salina lethality values were defined according to Clarkson's toxicity 43. After 24 hours of exposure to the control medium, all nauplii in the negative control group survived. In contrast, the positive control has an LC50 of 13.45 µg · mL-1, indicating high toxicity. Regarding the terms in Table 5, these are directly related to the LC50 and are inversely proportional, since, an extract or substance is potentially useful as highly toxic when its LC50 is ≤ 100 µg∙mL-1 (as accepted by the Clarkson Toxicity Index), and is considered very active; at concentrations between 100 µg∙mL-1 and 500 µg∙mL-1 it is considered medium toxic, with LC50 between 500 µg∙mL-1 and 1000 µg∙mL-1 it is considered low toxic, and with LC50 > 1000 µg∙mL-1 it would non-toxic, it would not be active; therefore, the higher the LC50, the lower the acute toxicity.43
The concentrations of KE, LE, and ME extracts showed moderate toxic activity (LC50: 100–500 µg mL-1) against A. salina nauplii, with LC50 values of 245.20, 261.27, and 151.40 µg∙mL⁻¹, respectively. Variation in toxicity is attributable to differences among plant species and varieties, resulting in qualitative and quantitative differences in the phytochemical content of primary and secondary metabolites in the extract.44 However, the observed biological effect is within the same classification of medium toxicity; this may be due to the common metabolites in the three extracts: coumarins and polyphenols.
The result obtained for the extract of K. pinnata (LC50 245.20 µg∙mL-1) is more representative than that reported in 2020, where the extract of this plant was evaluated, indicating an LC50 of 635 µg•mL-1, which is less active and higher than the value obtained in this research.45 Similarly, in a 2021 study on the essential oil of L. camara leaves, results report high toxicity, with LC50 values of 15.52 µg∙mL-1 against nauplii and 10.67 µg∙mL-1 against T47-D breast cancer cells.46
Another study on the fruits of M. azedarach reported moderate cytotoxicity, with an LC50 of 147.813 µg·mL-1. It is important to note that it is already known that the fruits of this plant contain tetranortriterpenoids. Based on these studies and the assay results, the cytotoxic potential of the plant extracts is evident.
Therefore, the biological activity observed in this research is closely related to the presence of the detected secondary metabolite families, which may act in complementary or synergistic ways when positive results are obtained, or counteract their effects (antagonistic effect) when negative results are obtained.
Preliminary studies indicate that the detected phytochemical compounds are important for various applications, including coumarins used in medical treatments for their photoreactive properties. They are also used as an insecticide, in addition to the other biological activities, such as the toxic effect on Artemia salina.48
In addition, some alkaloids are recognized for their antibacterial properties; likewise, phenolic compounds exhibit antioxidant effects and have highly antiseptic and bactericidal activity. Sterols are important in phytotherapy because they can lower cholesterol by reducing intestinal absorption. Finally, saponins with surfactant and hemolytic properties, both attributed to their structural characteristics of amphipathic nature, can also exert a wide range of biological and pharmacological activity, including insecticidal, antiprotozoal, anti-inflammatory, leishmanicidal, antitrichomonas, antiplatelet, broncholytic, hypocholesterolemic, as well as cytotoxic activity against various neoplasms.50
CONCLUSIONS
The phytochemical profile and antibacterial and cytotoxic activities of the ethanolic extracts from the leaves of these plants were evaluated. Preliminary chemical analysis of each ethanolic extract from the leaves of K. pinnata, L. camara, and M. azedarach showed the possible presence of sterols, coumarins, alkaloids, polyphenols, and saponins. The cytotoxicity test against A. salina nauplii showed that the ethanolic extracts of K. pinnata, L. camara, and M. azedarach leaves exhibited cytotoxicity, with LC50 values of 245.20, 261.27, and 151.40 µg·mL-1, respectively. Bioassays and chemical analyses indicate that the plants studied are potential sources of polar compounds with biologically active properties, which can be exploited in future research.
Funding Statement: This research received no external funding.
Conflict of Interest Statement: The authors declare no conflict of interest.
Author Contributions: Conceptualization, S.M.M. and Y.R.C.; methodology, Y.R.C. and H.H.M.; software, V.M.C. and H.R.C.; validation, H.H.M. and S.M.M.; formal analysis, Y.R.C. and V.M.C.; investigation, Y.R.C.; resources, S.M.M.; data curation, H.H.M.; writing—original draft preparation, Y.R.C.; writing—review and editing, S.M.M.; visualization, H.R.C.; supervision, V.M.C.; project administration, S.M.M.; funding acquisition, S.M.M. and Y.R.C.
Acknowledgments: The authors thank Yachay Tech Medicinal Chemistry Research Group (MedChem-YT) for institutional support.
Data Availability Statement: The data generated and/or analyzed during this study are available from the corresponding author upon reasonable request.
Institutional Review Board Statement: Not applicable.
Informed Consent Statement: Not applicable.
AI-Assisted Tools Disclosure: No artificial intelligence system was used to generate, manipulate, or analyze experimental data, images, or statistical outputs in this study. Generative AI tools were used exclusively for minor linguistic refinement and formatting standardization of the manuscript, under full human supervision. The authors independently verified all results, analyses, and conclusions in accordance with BioNatura Journal's policy: https://bionaturajournal.com/artificial-intelligence--ai-.html
REFERENCES
1. Forni C, Facchiano F, Bartoli M, Pieretti S, Facchiano A, D’Arcangelo D, et al. Beneficial role of phytochemicals on oxidative stress and age-related diseases. Biomed Res Int. 2019;2019:8748253. doi:10.1155/2019/8748253.
2. Sáenz DS. Drogas, fármacos y venenos. Rev Investig Educ Cienc Salud (RIECS). 2022;7(1):107-108. doi:10.37536/RIECS.2022.7.1.309.
3. Marchev AS, Yordanova ZP, Georgiev MI. Green (cell) factories for advanced production of plant secondary metabolites. Crit Rev Biotechnol. 2020;40(4):443-458. doi:10.1080/07388551.2020.1731414.
4. Newman DJ, Cragg GM. Natural products as sources of new drugs from 1981 to 2014. J Nat Prod. 2016;79(3):629-661. doi:10.1021/acs.jnatprod.5b01055.
5. Calderón-Chiu C, Calderón-Santoyo M, Ragazzo-Sánchez JA. Subproductos del cultivo de yaca en México: fuente de metabolitos secundarios y proteína. Ciencia y Sociedad. 2024;2(2):183-196. doi:10.5281/zenodo.12773852.
6. Pérez-Bautista I, Vilchis-Mercado I, Galaviz-Mauricio DC, Martínez-Betancourt SR, Sánchez-Sánchez ZS, Sánchez-Cortez K, et al. Desarrollos tecnológicos para el aprovechamiento industrial y farmacológico de la contrayerba (Pentalinon andrieuxii (Müll. Arg.) Hansen & Wunderlin). Agro-Divulgación. 2023;3(3). doi:10.54767/ad.v3i3.215.
7. Bello Pulido J, Franco-Salazar V, Vásquez-Suárez A. Florística de tres afloramientos geológicos y sus adyacencias en el extremo occidental de la Península de Araya, Venezuela. Saber. 2020;32.
8. Bello J. Plantas medicinales silvestres y/o naturalizadas en la península de Araya, Estado Sucre, Venezuela. Saber. 2017;29:326-339.
9. Jamilatun M, Kholisna M, Yulianto S. Physical quality and antifungal activity of Kalanchoe pinnata leaf extract ointment against Candida albicans. Quantum J Med Health Sci. 2024;3(5):18-26.
10. Saeed K, Chughtai MFC, Khaliq A, Liaqat A, Mehmood T, Khalid MZ, et al. Impact of extraction techniques and process optimization on antioxidant and antibacterial potential of Kalanchoe pinnata leaf extract. Int J Food Prop. 2024;27(1):909-926. doi:10.1080/10942912.2024.2373796.
11. Faundes-Gandolfo N, Jara-Gutiérrez C, Párraga M, Montenegro I, Vera W, Escobar M, et al. Kalanchoe pinnata (Lam.) Pers. leaf ethanolic extract exerts selective anticancer activity through ROS-induced apoptotic cell death in human cancer cell lines. BMC Complement Med Ther. 2024;24(1):269. doi:10.1186/s12906-024-04570-7.
12. Aisha K, Visakh NU, Pathrose B, Mori N, Baeshen RS, Shawer R. Extraction, chemical composition and insecticidal activities of Lantana camara Linn. leaf essential oils against Tribolium castaneum, Lasioderma serricorne and Callosobruchus chinensis. Molecules. 2024;29(2):344. doi:10.3390/molecules29020344.
13. Patel K, Patil M, Abhale Y, Dateer RB, Kumar D, Larios AP, et al. A critical review on Lantana camara extract-mediated bio-inspired synthesis of nanomaterials and their potential application: challenges and future perspectives. Inorg Chem Commun. 2025;173:113830. doi:10.1016/j.inoche.2024.113830.
14. Pérez-Galicia D, Palma-Lara I, Espinosa-Aguirre JJ. Ecotoxicological evaluation of an aqueous phytoextract of Melia azedarach L. Sci Total Environ. 2024;950:175314. doi:10.1016/j.scitotenv.2024.175314.
15. Touzout SN, Merghni A, Laouani A, Boukhibar H, Alenazy R, Alobaid A, et al. Antibacterial properties of methanolic leaf extracts of Melia azedarach L. against gram-positive and gram-negative pathogenic bacteria. Microorganisms. 2023;11(8):2062. doi:10.3390/microorganisms11082062.
16. Marcano D, Hasegawa M. Fitoquímica orgánica. 2a ed. Caracas: Universidad Central de Venezuela, Consejo de Desarrollo Científico y Humanístico; 2002.
17. Murillo E, Méndez J. Guía metodológica para la detección rápida de algunos metabolitos secundarios. Ibagué: Departamento de Química, Facultad de Ciencias, Universidad del Tolima; 2007.
18. Bauer AW, Kirby WM, Sherris JC, Turck M. Antibiotic susceptibility testing by a standardized single disk method. Am J Clin Pathol. 1966;45(4):493-496.
19. Meyer BN, Ferrigni NR, Putnam JE, Jacobsen LB, Nichols DE, McLaughlin JL. Brine shrimp: a convenient general bioassay for active plant constituents. Planta Med. 1982;45:31-34. doi:10.1055/s-2007-971236.
20. Ramos Otiniano CC, Lezama Asencio PB, Hidalgo Rodríguez JEM. Actividad biocida de extractos de hojas y semillas de Melia azedarach (Meliaceae) sobre larvas de Spodoptera frugiperda (Lepidoptera: Noctuidae). Arnaldoa. 2022;29(3):427-438. doi:10.22497/arnaldoa.293.29304.
21. Cortez PM. Análisis de los espectros de infrarrojo. In: Principios y aplicaciones de la espectroscopia de infrarrojo en el análisis de alimentos y bebidas. 2020. p. 66-82.
22. Bunaciu AA, Aboul-Enein HY, Fleschin S. Recent applications of Fourier transform infrared spectrophotometry in herbal medicine analysis. Appl Spectrosc Rev. 2011;46(4):251-260. doi:10.1080/05704928.2011.565532.
23. Rubinson KA, Rubinson JF. Análisis instrumental. Madrid: Pearson Educación; 2000.
24. Moreno S, Hernández J, Nuñez J. Metabolitos bioactivos de la culebra ciega (Anguis fragilis). Braz J Anim Environ Res. 2023;6(4):3484-3497. doi:10.34188/bjaerv6n4-032.
25. Guillén-Andrade H, Escalera-Ordaz AK, Torres-Gurrola G, García-Rodríguez YM, Espinosa García FJ, Tapia-Vargas LM. Identificación de nuevos metabolitos secundarios en Persea americana Miller variedad Drymifolia. Rev Mex Cienc Agríc. 2019;10(SPE23):253-265. doi:10.29312/remexca.v0i23.2025.
26. Castillo ÓC, Mora GC, Martínez IR. Identificación de alcaloides en corteza de Uncaria tomentosa y discusión sobre su potencial farmacológico. InvestFarma. 2021;1(1).
27. Sousa JSP, França JP, França LP, Chaves ALF, Conceição AO. Caracterização histoquímica de órgãos vegetativos de Lantana camara L. (Verbenaceae) ocorrente no sul da Bahia. Braz J Dev. 2020;6(8):63840-63848. doi:10.34117/bjdv6n8-711.
28. Ayala-Mata F, Barrera-Mendoza CC, Cortés-Rojo C, Montoya-Pérez RDC, García-Pérez ME, Rodríguez-Orozco AR. Antioxidantes en asma: polifenoles. Med Interna Mex. 2019;35(2):223-234. doi:10.24245/mim.v35i2.2291.
29. Navarrete Barragán NA, Pita-Ospina EF, Sánchez Mora RM, Giraldo Quintero SE, Bernal Lizarazú MC. Actividad in vitro de los extractos etanólicos de Lantana camara L., Petiveria alliacea L. y Lippia dulcis T. frente a bacterias patógenas. Nova. 2020;18(33):53-71. doi:10.22490/24629448.3700.
30. Nava Álvarez R, Juárez Juárez M, Bolaños Valerio E, Morales Jaimes M. Determinación de cumarinas con actividad antioxidante por cromatografía de líquidos de alta resolución. 2015.
31. Castro Mandujano ON, Álvarez Bautista JL. Análisis de ácidos grasos y esteroles del Líquen Everniopsis trulla. Rev Boliv Quím. 2021;38(3):104-112.
32. Moreno LGN, Agosto JG, Hipólito CN. Composición fitoquímica y propiedades antioxidantes de la planta mala madre (Kalanchoe pinnata). South Flor J Dev. 2023;4(1):201-214. doi:10.46932/sfjdv4n1-014.
33. Shrestha SS, Ferrarese I, Sut S, Zengin G, Grana S, Ak G, et al. Phytochemical investigations and in vitro bioactivity screening on Melia azedarach L. leaves extract from Nepal. Chem Biodivers. 2021;18(5):e202001070. doi:10.1002/cbdv.202001070.
34. Loor JDB, Boza FAA, Mendoza ALC, Vidal LRL, Marcillo GAC. Fenoles simples provenientes de la destilación de Origanum vulgare evaluados por espectrometría UV utilizando auxocromos. Rev Inst Investig Fac Minas Metal Cienc Geogr. 2021;24(48):189-195.
35. Silverstein RM, Webster FX, Kiemle DJ. Spectrometric identification of organic compounds. 7th ed. New York: John Wiley & Sons; 2005.
36. Giono-Cerezo S, Santos-Preciado JI, Rayo Morfín-Otero MD, Torres-López FJ, Alcántar-Curiel MD. Resistencia antimicrobiana. Importancia y esfuerzos por contenerla. Gac Med Mex. 2020;156(2):172-180. doi:10.24875/GMM.20005624.
37. Quirós Fernández LM, Vázquez F, Fernández-Vega I, Rodríguez D, Martín C. Edición de un libro de divulgación científica sobre revisiones de actualidad en temas de microbiología sanitaria. 2022.
38. Rivero-Pérez N, Prieto-Méndez J, Hernández-Fuentes A, Zaragoza-Bastida A, Madariaga-Navarrete A. Efecto antihelmíntico y antibacteriano in vitro del extracto hidroalcohólico de hojas y tallos de Kalanchoe daigremontiana. Abanico Vet. 2022;12:102:1-16. doi:10.21929/abavet2022.1.
39. Chanda S, Baravalia Y. Brine shrimp cytotoxicity of Caesalpinia pulcherrima aerial parts, antimicrobial activity and characterisation of isolated active fractions. Nat Prod Res. 2011;25(20):1955-1964. doi:10.1080/14786419.2010.530600.
40. Baravalia Y, Vaghasiya Y, Chanda S. Brine shrimp cytotoxicity, anti-inflammatory and analgesic properties of Woodfordia fruticosa Kurz flowers. Iran J Pharm Res. 2012;11(3):851-861.
41. Pertiwi D, Hafiz I, Leny. Potential bioactivities of ethanol, ethyl acetate and n-hexane extracts from pagoda leaves (Clerodendrum paniculatum L.). Rasayan J Chem. 2020;13(4):2313-2316. doi:10.31788/RJC.2020.1345791.
42. Yoshida T, Chou T, Nitta A, Miyamoto KI, Koshiura R, Okuda T. Woodfordin C, a macro-ring hydrolyzable tannin dimer with antitumor activity, and accompanying dimers from Woodfordia fruticosa flowers. Chem Pharm Bull (Tokyo). 1990;38(5):1211-1217. doi:10.1248/cpb.38.1211.
43. Clarkson C, Maharaj VJ, Crouch NR, Grace OM, Pillay P, Matsabisa MG, et al. In vitro antiplasmodial activity of medicinal plants native to or naturalised in South Africa. J Ethnopharmacol. 2004;92(2-3):177-191. doi:10.1016/j.jep.2004.02.011.
44. Ni YW, Lin KH, Chen KH, Wu CW, Chang YS. Flavonoid compounds and photosynthesis in Passiflora plant leaves under varying light intensities. Plants (Basel). 2020;9(5):633. doi:10.3390/plants9050633.
45. Coe FG, Parikh DM, Johnson CA, Kucharczyk M, Tovar JA. Bioactivity of 68 species of medicinal plants of eastern Nicaragua. J Pharmacogn Phytochem. 2020;9(3):101-112.
46. Suryati S, Aziz ED, Efdi M, Wahyuni FS, Hefni D. Analysis of the essential oil from Lantana camara leaves and its cytotoxic potential against T-47D breast cancer cells. J Ris Kim. 2021;12(1):1-9. doi:10.25077/jrk.v12i1.364.
47. Sadaf HM, Bibi Y, Arshad M, Razzaq A, Ahmad S, Iriti M, et al. Analysis of Peganum harmala, Melia azedarach and Morus alba extracts against six lethal human cancer cells and oxidative stress along with chemical characterization through advance Fourier transform and nuclear magnetic resonance spectroscopic methods towards green chemotherapeutic agents. Saudi Pharm J. 2021;29(6):552-565. doi:10.1016/j.jsps.2021.04.016.
48. Menezes ACN, de Oliveira AKM, Roel AR, de Andrade Porto KR, Favero S, Matias R. Sapindus saponaria (Sapindaceae): chemical composition and toxic effect on Artemia salina (Artemiidae) and Aedes aegypti (Culicidae). Rev Gest Soc Ambient. 2023;17(10):e03942. doi:10.24857/rgsa.v17n10-016.
49. Nerdy N, Lestari P, Sinaga JP, Ginting S, Zebua NF, Mierza V, et al. Brine shrimp (Artemia salina Leach.) lethality test of ethanolic extract from green betel (Piper betle Linn.) and red betel (Piper crocatum Ruiz & Pav.) through the soxhletation method for cytotoxicity test. Open Access Maced J Med Sci. 2021;9(A):407-412. doi:10.3889/oamjms.2021.6171.
50. Nur S, Mubarak F, Jannah C, Winarni DA, Rahman DA, Hamdayani LA, et al. Total phenolic and flavonoid compounds, antioxidant and toxicity profile of extract and fractions of paku atai tuber (Angiopteris ferox Copel). Food Res. 2019;3(6):734-740. doi:10.26656/fr.2017.3(6).135.
51. Musdalipah M, Tee SA, Karmilah K, Sahidin S, Fristiohady A, Yodha AWM. Total phenolic and flavonoid content, antioxidant, and toxicity test with BSLT of Meistera chinensis fruit fraction from Southeast Sulawesi. Borneo J Pharm. 2021;4(1):6-15. doi:10.33084/bjop.v4i1.1686.
52. Macías Villamizar VE, Badrán Padilla R, Rodríguez Ochoa J, Silva Román H. Metabolitos secundarios, actividad biológica y etnobotánica de plantas de Santa Marta. Santa Marta: Editorial Unimagdalena; 2021.
Received: November 30, 2025 / Accepted: January 13, 2026 / Published (online): March 15, 2026 (Europe/Madrid)
Citation. Moreno Morales S, Rivas Castañeda Y, Herrera Mata H, Morera Córdova V, Rodríguez Cabrera H. Phytochemical evaluation and biological activity of three plant extracts from Cumaná, Venezuela. BioNatura Journal: Ibero-American Journal of Biotechnology and Life Sciences. 2026;3(1):4. https://doi.org/10.70099/BJ/2026.03.01.4
Correspondence should be addressed to: smoreno@yachaytech.edu.ec
Peer Review Information BioNatura Journal thanks the anonymous reviewers for their valuable contribution to the peer-review process. Regional peer-review coordination was conducted under the BioNatura Institutional Publishing Consortium (BIPC), involving:
• Universidad Nacional Autónoma de Honduras (UNAH)
• Universidad de Panamá (UP)
• RELATIC (Panama)
Reviewer selection and assignment were supported via: https://reviewerlocator.webofscience.com/
• Universidad Nacional Autónoma de Honduras (UNAH)
• Universidad de Panamá (UP)
• RELATIC (Panama)
Reviewer selection and assignment were supported via: https://reviewerlocator.webofscience.com/
Publisher Information Published by Clinical Biotec S.L. (Madrid, Spain) as the publisher of record under the BioNatura Institutional Publishing Consortium (BIPC). Places of publication: Madrid (Spain); Tegucigalpa (Honduras); Panama City (Panama). Online ISSN: 3020-7886.
Open Access Statement All articles published in BioNatura Journal are freely and permanently available online immediately upon publication, without subscription charges or registration barriers.
Publisher's Note BioNatura Journal remains neutral regarding jurisdictional claims in published maps and institutional affiliations.
Copyright and License © 2026 by the authors. This article is published under the terms of the Creative Commons Attribution (CC BY 4.0) license, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
License details: https://creativecommons.org/licenses/by/4.0/
License details: https://creativecommons.org/licenses/by/4.0/
Governance For editorial governance and co-publisher responsibilities, see the BIPC Governance Framework (PDF) at: https://clinicalbiotec.com/bipc
