Personalized treatment with Fresh Frozen Plasma for the pro-inflammatory septic phenotype characterized by refractory shock, hemodynamic incoherence, and endothelial damage. A proof of concept
1. Hospital Pablo Arturo Suárez, Unidad de Terapia Intensiva, Centro de Investigación Clínica, Quito,Ecuador
2. Universidad Central del Ecuador, Facultad de Ciencias Médicas, Escuela de Medicina, Cátedra de Medicina Crítica e Inmunología
*Corresponding Author: Jorge Luis Vélez-Páez, Email: jlvelez@uce.edu.ec
ABSTRACT
This prospective, single-center observational study evaluated the association between early (within the first 24 hours) administration of fresh-frozen plasma (FFP) and clinical outcomes in 30 patients with catecholamine-resistant septic shock, a pro-inflammatory phenotype, and endothelial damage. Patients who received FFP had greater baseline severity, reflected in higher lactate concentrations on admission (5.12 vs. 2.10 mmol/L; p = 0.005), higher initial norepinephrine (NE) doses (0.30 vs. 0.18 µg/kg/min; p = 0.022), and greater use of a second vasopressor (56% vs. 4.8%; p = 0.005). This result suggests that FFP was preferentially used as rescue therapy in patients with greater hemodynamic and metabolic severity. Despite this, vasopressor support showed a progressive reduction in both groups, with significant differences between groups at 24 hours (FFP/control ratio 1.93; p = 0.019), followed by convergence and no further divergence at 48 and 72 hours. Fresh frozen plasma (FFP) demonstrated an acceptable safety profile and early hemodynamic changes, but did not significantly reduce ICU mortality compared to conventional therapy. These findings suggest that, while FFP may have modulating potential, its clinical efficacy remains open to further investigation and warrants larger randomized controlled trials to determine optimal dosage and duration of therapy.
Keywords: septic shock; fresh frozen plasma; hemodynamic incoherence; vasopressors; critical care.
INTRODUCTION
Septic shock is the most severe form of sepsis and is defined by persistent hypotension requiring vasopressors to maintain a mean arterial pressure ≥65 mmHg, along with serum lactate levels >2 mmol/L despite adequate fluid resuscitation, a condition associated with in-hospital mortality exceeding 40%1,2. Although norepinephrine (NE) is the first-line vasopressor, a significant subgroup of patients progresses to catecholamine-resistant septic shock, with persistent tissue hypoperfusion and organ dysfunction despite normalization of macrocirculatory parameters3. This uncoupling is largely explained by hemodynamic incoherence, a phenomenon characterized by the dissociation between the restoration of blood pressure or cardiac output and the persistence of microcirculatory dysfunction, ultimately responsible for oxygen transport to tissues 4–6. Hemodynamic incoherence is common in septic shock and is associated with the progression of multiple organ failure7.
Sepsis is a heterogeneous syndrome resulting from a profoundly dysregulated immune and vascular response to infection, rather than from the direct effect of the pathogen. Pro-inflammatory and prothrombotic phenotypes, driven by cytokines such as interleukin-6, tumor necrosis factor, and IL-1β, are associated with activation of coagulation, inhibition of fibrinolysis, and endothelial dysfunction, causing clinical variability and divergent therapeutic responses8,9. Clinical and molecular studies have demonstrated that these subphenotypes exhibit distinct risks and responses to treatment, helping explain the failure of uniform medical strategies in sepsis and shock10. A central component of this pathophysiology is damage to the endothelial glycocalyx, whose degradation leads to increased vascular permeability, capillary leakage, complement activation, and microvascular thrombosis. Biomarkers such as syndecan-1 reflect this damage and are consistently associated with greater severity, vasopressor requirements, and mortality11.
In this context of hemodynamic incoherence, biological heterogeneity, and endothelial dysfunction, adjuvant therapies such as fresh frozen plasma (FFP) and plasma exchange have emerged, with the capacity to modulate the inflammatory response, restore protective plasma components, and improve vascular integrity. However, the available clinical evidence is limited and heterogeneous, stressing the need to evaluate their impact on relevant clinical outcomes in actual practice settings and to generate future hypotheses that more strongly support their use.
MATERIAL AND METHODS
Study Design and Setting
A prospective, observational study was conducted in the Intensive Care Unit (ICU) of Hospital Pablo Arturo Suárez in Quito, Ecuador. Patients were followed from admission until discharge or death in the ICU. The decision to administer FFP or conventional therapy (CT) was made exclusively by the attending physician, without investigator intervention.
Population and Selection Criteria
Adult patients (≥18 years) diagnosed with septic shock according to Sepsis-3 criteria were included using consecutive non-probability sampling. Inclusion criteria included at least 24 hours of shock duration, a surgically resolved or clinically resolving infectious focus, a NE requirement between 0.1 and 0.3 µg/kg/min, and elevated inflammatory markers defined as ferritin ≥750 ng/mL and/or interleukin-6 (IL-6) ≥500 pg/mL. Eligibility required meeting at least one of these inflammatory criteria; in patients from the conventional therapy group, inclusion was predominantly based on elevated ferritin levels, even when IL-6 values were below the predefined threshold. Patients with prior use of a second vasoactive agent, transfusion for other clinical indications, documented limitations on therapeutic effort, pregnancy, or refusal of the informed consent were excluded.
Study Groups and Variables
Patients were classified into two groups based on the treatment received: CT or FFP. Demographic variables, clinical severity (lactate and SOFA score), vasopressor requirements, need for mechanical ventilation, cumulative fluid balance at 24, 48, and 72 hours, and at ICU discharge, inflammatory biomarkers (ferritin, interleukin-6, and D-dimer), as well as the clinical outcomes of mortality and length of stay in the ICU, were recorded.
Procedure and Data Collection
Patients were identified daily by reviewing the ICU admission census. After verifying compliance with the selection criteria and acquiring informed consent, prospective and systematic collection of clinical, hemodynamic, and laboratory variables was performed. NE doses were recorded upon admission and longitudinally at 24, 48, and 72 hours. Each patient was identified using an alphanumeric code to ensure data anonymization and traceability.
Fresh frozen plasma administration (exposure)
FFP was administered as part of routine clinical care at the attending intensivist's discretion. The local pragmatic regimen consisted of one unit of FFP (approximately 150 mL) every 8 hours for 24 hours (i.e., three administrations, total volume approximately 450 mL per patient). The first dose was initiated as soon as the predefined inclusion criteria were met, including persistent vasopressor requirement and elevated inflammatory markers. Infusion duration or rate was not systematically recorded; however, FFP was administered according to institutional transfusion practice. The clinical rationale for FFP administration was persistent catecholamine requirement despite standard therapy, in a pragmatic real-world context.
Safety monitoring.
Patients were actively monitored for transfusion-related adverse events, including transfusion-related acute lung injury (TRALI) and transfusion-associated circulatory overload (TACO), during and up to 24 hours after each FFP administration. TRALI was defined as new acute hypoxemia with bilateral pulmonary infiltrates not attributable to cardiac failure or fluid overload, and TACO as acute respiratory distress with clinical evidence of volume overload requiring intervention. When clinically indicated, bedside transthoracic echocardiography was performed as part of routine ICU care to assess ventricular function, volume status, and signs of acute cardiac dysfunction or fluid intolerance. Adverse events were prospectively recorded in the clinical chart and reviewed daily by the study team.
Concomitant therapies
All patients received standard care for septic shock according to international guidelines, including early antimicrobial therapy, source control, fluid resuscitation, and norepinephrine as first-line vasopressor. Additional supportive therapies, such as mechanical ventilation, renal replacement therapy, and corticosteroids, were prescribed at the attending physician's discretion based on clinical indications. Epinephrine was used as the second-line vasopressor when required, in accordance with local ICU protocols, given the unavailability of vasopressin in our setting. These cointerventions were not protocolized and were not used as criteria for group allocation.
Statistical analysis
Statistical analysis was performed using R software version 4.5.3, with p-values < 0.05 considered statistically significant. The normality of quantitative variables was assessed using the Shapiro-Wilk test; those with a non-normal distribution were described as median and interquartile range (P25–P75). Qualitative variables were expressed as absolute frequencies and percentages. Bivariate comparisons between groups were performed using the Mann-Whitney U test for continuous variables and the chi-square test or Fisher's exact test for categorical variables. Longitudinal analysis of NE requirement was performed using a generalized linear mixed model with a Gamma distribution and log linkage, including group, time, and their interaction as fixed effects, and an intercept per patient as a random effect. Model adequacy was assessed by graphical inspection of the residuals.
Ethical Considerations
The study was approved by the Research Ethics Committee of the Eugenio Espejo Hospital (CEISH-HEEE-2025-021) as minimal-risk research. Data were anonymized using alphanumeric coding, preserving confidentiality and compliance with the principles of the Declaration of Helsinki.
RESULTS
Thirty patients were included: 9 in the FFP group and 21 in the conventional therapy (CT) group.
Age was similar between groups. The sex distribution did not differ (male: 67% in FFP vs. 48% in CT; p = 0.440). The frequency of diabetes and hypertension was similar, as was BMI in the FFP vs. CT group comparison. (Table 1)
Characteristic
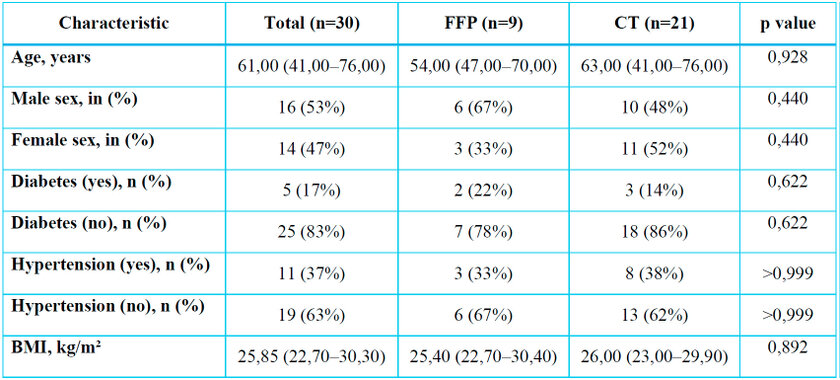
Data as median (Q1–Q3) or n (%). Wilcoxon/Fisher test as appropriate.
FFP: fresh frozen plasma; BMI: body mass index.
Table 1. Demographic characteristics and comorbidities (FFP vs. CT)
No differences were found between groups in ferritin at admission, IL-6, or D-dimer. Leukocytes, neutrophils, platelets, and MPV did not differ between groups. (Table 2.)
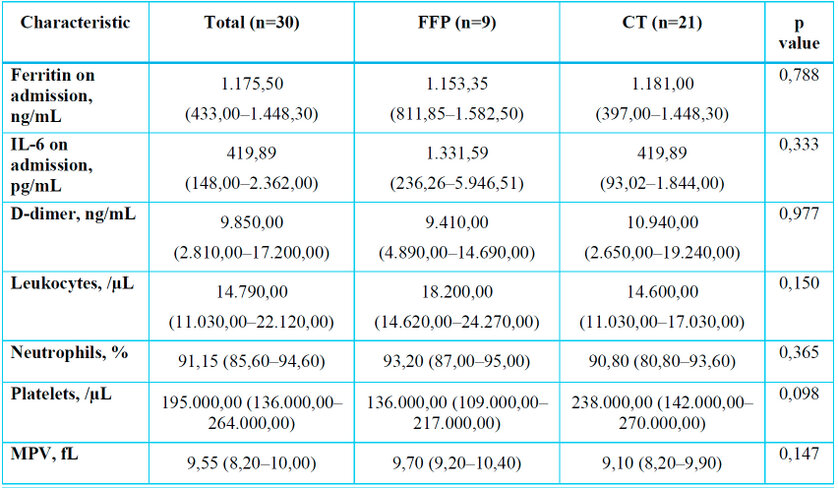
FFP: fresh frozen plasma; IL-6: interleukin-6; MPV: mean platelet volume.
Table 2. Analytical and inflammatory parameters (FFP vs. conventional therapy)
In the first 24 hours, the maximum vasoactive dose was higher in the FFP group, with a median of 0.30 (0.23–0.63), compared with 0.18 (0.10–0.30) in the CT group, a difference that was statistically significant (p = 0.022). No significant differences in the maximum vasoactive dose were observed at 48 or 72 hours. At the start of FFP in the plasma group, the median NE dose was 0.38 (0.26–0.45). (Table 3)
The use of a second vasoactive agent was more frequent in the FFP group (56%) than in the CT group (4.8%), with a statistically significant difference (p = 0.005). Among those who received a second vasoactive agent, epinephrine was used in 100% of cases in both groups, with no difference (p > 0.999). No differences were observed in fluid balance at 24, 48, and 72 hours, or at discharge (Table 3).
ICU mortality was similar between groups (FFP: 11% vs. CT: 24%; p = 0.637). However, ICU length of stay was longer in the FFP group, with a median of 7.0 (6.0–9.0) days compared to 4.0 (3.0–6.0) days in the conventional therapy group (p = 0.042). The proportion of patients requiring mechanical ventilation did not differ between groups (67% in FFP vs. 48% in CT; p = 0.440). (Table 3)
The SOFA score on admission was comparable (FFP: 10.00 [8.00–13.00] vs. CT: 10.00 [8.00–12.00]; p = 0.964). At discharge, the SOFA score tended to be higher in FFP (4.00 [2.00–6.00]) compared to CT (2.00 [1.00–6.00]), although this difference did not reach statistical significance (p = 0.102).
Lactate levels on admission were significantly higher in the FFP group than in the CT group (p = 0.005). Central venous oxygen saturation was similar between groups. Capillary refill on admission was prolonged in both groups (FFP: 4.00 [4.00–5.00] vs conventional therapy: 3.00 [3.00–4.00]; p = 0.223), with no significant difference. (Table 3)
No TRALI or TACO events were observed in this sample.
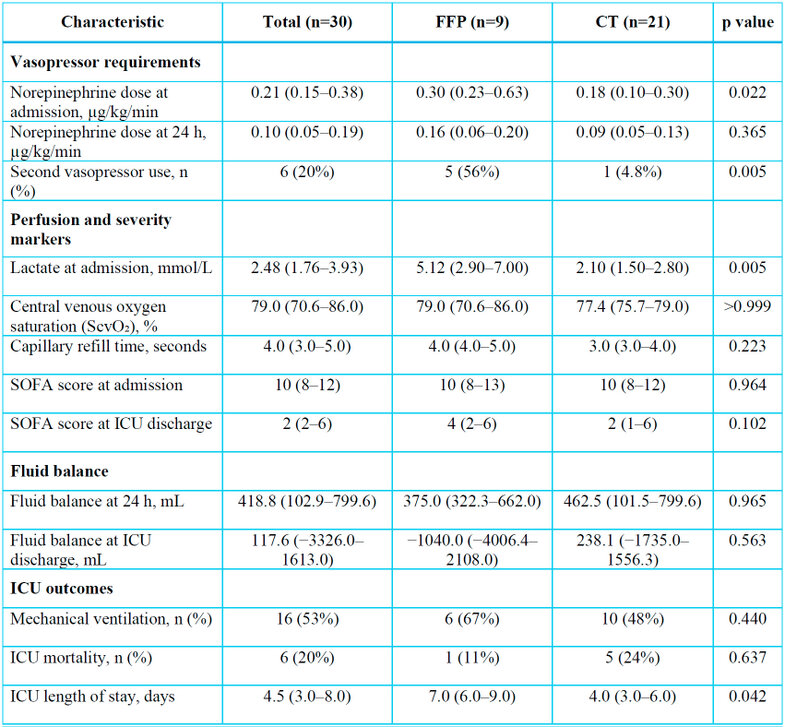
Data are presented as median (interquartile range) or n (%). Comparisons were performed using the Mann–Whitney U test or Fisher's exact test, as appropriate.
FFP: fresh frozen plasma; CT: conventional therapy; ICU: intensive care unit; SOFA: Sequential Organ Failure Assessment; ScvO₂: central venous oxygen saturation.
Table 3. Hemodynamic variables, perfusion parameters, and ICU outcomes.
Longitudinal analysis of vasoactive agents (GLMM Gamma)
Estimated marginal measures (EMMs) showed a decrease in vasoactive support over time in both groups. At 24 hours, the FFP group had higher values than conventional therapy, with an FFP-to-conventional therapy ratio of 1.93 and statistical significance (p = 0.019). At 48 hours, no difference was observed between groups (p = 0.377), and at 72 hours, no conclusive difference was documented either (p = 0.412), with a trend towards less support in FFP. (Figure 1)
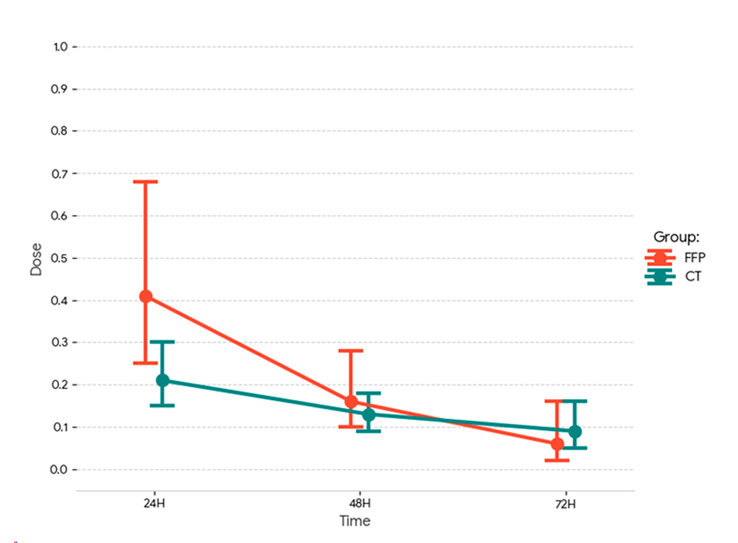
Figure 1. Estimated marginal means of norepinephrine dose (µg/kg/min) at 24, 48, and 72 hours in the FFP and conventional therapy groups. Error bars indicate 95% confidence intervals.
In within-group comparisons, both groups showed significant decreases from 24 hours onward. In the FFP group, the reduction was observed between 24 and 48 hours (p = 0.014) and between 24 and 72 hours (p = 0.001). In the conventional therapy group, significant decreases were also observed between 24 and 48 hours (p = 0.049) and between 24 and 72 hours (p = 0.016). Comparisons between 48 and 72 hours showed no significant differences in either group.
Group*Time interactions did not reach significance at the 5% level, indicating limited evidence that the time trajectory differs between strategies, although an inconclusive signal was observed at 72 hours (p = 0.073). Overall, the results support the finding that the difference between groups is concentrated at the 24-hour point, with no statistically significant differences at 48 and 72 hours.
DISCUSSION
In this prospective study, patients who received FFP presented with greater baseline severity, reflected by higher lactate concentrations at admission (5.12 vs. 2.10 mmol/L; p = 0.005), higher initial doses of NE (0.30 vs. 0.18 µg/kg/min; p = 0.022), and greater use of a second vasopressor (56% vs. 4.8%; p = 0.005). This result suggests that FFP was preferentially used as a rescue therapy in patients with greater hemodynamic and metabolic severity. Consequently, the observed differences must be interpreted in the context of a high risk of confounding by indication, which precludes causal inference regarding the effects of FFP. Despite this, the evolution of vasopressor support showed a progressive reduction in both groups, with significant differences between groups at 24 hours (FFP/control ratio 1.93; p = 0.019), followed by convergence and no subsequent divergences at 48 and 72 hours.
This pattern is clinically relevant, since it suggests that, even in patients with greater initial compromise, the use of FFP was associated with initial and subsequently sustained hemodynamic improvement compared to the control group. In the context of septic shock, the progressive increase in catecholamines not only indicates vasoplegia but also endothelial and microcirculatory dysfunction, phenomena that are not always corrected by normalization of blood pressure12,13. Multiple studies have reinforced the concept of hemodynamic incoherence, in which macrocirculatory stability does not guarantee adequate tissue perfusion, particularly in patients with severe inflammatory phenotypes4–7.
The physiological rationale for FFP in sepsis is its potential to modulate endothelial damage and glycocalyx degradation14. Recent experimental and translational evidence has shown that plasma can attenuate endothelial barrier disruption, reduce capillary extravasation, and modulate key inflammatory mediators, suggesting a possible early benefit for vascular stability 14–16. However, contemporary clinical studies in humans have shown heterogeneous results, with transient hemodynamic effects that do not consistently translate into reduced mortality. In cohorts of patients with sepsis and septic shock, early transfusion of FFP has not shown benefits in 28-, 30-, or 90-day mortality, in decreasing the need for fluids or vasopressors, or in the length of stay in the ICU or on mechanical ventilation 17,18.
In our cohort, no significant differences were observed between groups in ferritin, interleukin-6, or D-dimer levels at admission, showing a high and comparable inflammatory and prothrombotic burden in both groups. This finding is consistent with recent studies showing that conventional serum biomarkers may not adequately detect dynamic changes at the endothelial or microvascular levels induced by therapies targeting the plasma compartment19. The lack of direct assessment of endothelial glycocalyx damage using biomarkers such as syndecan-1 limits the physiological interpretation of FFP's potential effects on endothelial function. Given that glycocalyx degradation is associated with increased vascular permeability, endothelial dysfunction, and worse outcomes in shock and sepsis, this limitation prevents the establishment of conclusive mechanistic links between the intervention and the observed clinical changes 16,20.
Regarding clinical outcomes, no differences in ICU mortality were observed between groups (11% in the FFP group vs. 24% in the conventional therapy group; p = 0.637), nor in the proportion of patients requiring mechanical ventilation. These findings are consistent with contemporary observational evidence in septic shock/sepsis, where plasma transfusion has not been associated with a reproducible survival benefit after adjustment for baseline severity, despite a plausible mechanistic rationale and occasional early physiologic or endothelial/hemodynamic signals reported in the literature 21–23. Patients treated with FFP had a longer ICU length of stay (median 7 vs. 4 days; p = 0.042), which likely reflects confounding by indication—FFP tends to be administered preferentially to patients with greater baseline severity and clinical complexity—rather than a direct effect of the intervention itself21,24,25.
Importantly, no major transfusion-related complications were observed, including transfusion-related acute lung injury (TRALI) or transfusion-associated circulatory overload (TACO). This is compatible with the absence of significant differences in cumulative fluid balance between groups throughout the ICU stay, suggesting that FFP administration was not associated with clinically relevant volume overload in this cohort; nevertheless, active-surveillance meta-analyses indicate that TRALI and TACO, while uncommon, remain clinically relevant outcomes that require systematic detection and adequate sample size to be reliably assessed 26–28.
Several restrictions must be acknowledged. The observational and non-randomized design entails a substantial risk of confounding by indication, as FFP was preferentially administered to patients with greater baseline severity. In addition, the small sample size and imbalance between groups reduce statistical power and increase uncertainty around effect estimates, particularly in the FFP group. Finally, the single-center design may limit generalizability. Accordingly, these results should be interpreted as exploratory and hypothesis-generating rather than confirmatory, in line with reporting guidance for observational studies 29.
CONCLUSIONS
Overall, our results suggest that FFP use was associated with a clinically acceptable safety profile, with no evidence of fluid overload or serious transfusion events, and may exert an early hemodynamic effect in patients with catecholamine-resistant septic shock, hemodynamic incoherence, and a pro-inflammatory phenotype with endothelial damage. Our results should be considered preliminary evidence and a generating hypothesis, and multicenter studies with larger sample sizes, better control of confounding factors, and direct assessment of endothelial damage are needed to more precisely define the role of FFP, timing of administration, and dosage in the management of septic shock.
Funding Statement: This research received no external funding.
Conflict of Interest Statement: The authors declare no conflicts of interest.
Author Contributions: Conceptualization, J.V., E.O., F.C., and G.J.; methodology, J.V., E.O., F.C., and G.J.; formal analysis, J.V., E.O., F.C., and G.J.; statistical analysis, J.V., E.O., F.C., and G.J.; writing—original draft preparation, J.V., E.O., F.C., and G.J.; writing—review and editing, J.V., E.O., F.C., and G.J.; approval of the final version, J.V., E.O., F.C., and G.J.
Acknowledgments: The authors thank the medical and nursing staff of the Intensive Care Unit at Hospital Pablo Arturo Suárez for their support.
Data Availability Statement: The data generated and/or analyzed during this study are available from the corresponding author upon reasonable request.
Institutional Review Board Statement: The study was approved by the Research Ethics Committee of Eugenio Espejo Hospital (CEISH-HEEE-2025-021).
Informed Consent Statement: Informed consent was obtained from all subjects involved in the study.
AI-Assisted Tools Disclosure: No artificial intelligence system was used to generate, manipulate, or analyze experimental data, images, or statistical outputs in this study. Generative AI tools were used exclusively for minor linguistic refinement and formatting standardization of the manuscript, under full human supervision. The authors independently verified all results, analyses, and conclusions in accordance with BioNatura Journal's policy: https://bionaturajournal.com/artificial-intelligence--ai-.html
REFERENCIAS
1. Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):801-810. doi:10.1001/jama.2016.0287.
2. Shankar-Hari M, Phillips GS, Levy ML, Seymour CW, Liu VX, Deutschman CS, et al. Developing a New Definition and Assessing New Clinical Criteria for Septic Shock (Sepsis-3). JAMA. 2016;315(8):775-787. doi:10.1001/jama.2016.0289.
3. Leone M, Einav S, Antonucci E, Depret F, Lakbar I, Martin-Loeches I, et al. Multimodal strategy to counteract vasodilation in septic shock. Anaesth Crit Care Pain Med. 2023;42(3):101193. doi:10.1016/j.accpm.2023.101193.
4. Ince C. Hemodynamic coherence and the rationale for monitoring the microcirculation. Crit Care. 2015;19(Suppl 3):S8. doi:10.1186/cc14726.
5. Bakker J, Ince C. Monitoring coherence between the macro and microcirculation in septic shock. Curr Opin Crit Care. 2020;26(3):267-272. doi:10.1097/MCC.0000000000000729.
6. Páez JLV, Ochoa-Robles E. Modelo 4V-2C: Una visión nemotécnica de los fenotipos en la incoherencia hemodinámica. Rev Argent Ter Intensiva [Internet]. 2025;42(1). Disponible en: https://revista.sati.org.ar/index.php/MI/article/view/960/1171
7. Huang L, Huang Q, Ma W, Yang H. Understanding hemodynamic incoherence: mechanisms, phenotypes, and implications for treatment. Shock. 2025;63(3):342-350. doi:10.1097/SHK.0000000000002507.
8. Levi M, van der Poll T. Coagulation and sepsis. Thromb Res. 2017;149:38-44. doi:10.1016/j.thromres.2016.11.007.
9. Semeraro N, Ammollo CT, Semeraro F, Colucci M. Sepsis, thrombosis and organ dysfunction. Thromb Res. 2012;129(3):290-295.
10. Gordon AC, Alipanah-Lechner N, Bos LD, Dianti J, Diaz JV, Finfer S, et al. From ICU Syndromes to ICU Subphenotypes: Consensus Report and Recommendations for Developing Precision Medicine in the ICU. Am J Respir Crit Care Med. 2024;210(2):155-166. doi:10.1164/rccm.202311-2086SO.
11. Tang F, Zhao XL, Xu LY, Zhang JN, Ao H, Peng C. Endothelial dysfunction: Pathophysiology and therapeutic targets for sepsis-induced multiple organ dysfunction syndrome. Biomed Pharmacother. 2024;178:117180. doi:10.1016/j.biopha.2024.117180.
12. Sathianathan S, Sachar S, Berro J, Nero N, Bauer SR, Tonelli AR, et al. Vasoactive medications and the microcirculation in septic shock: a scoping review. Crit Care Med. 2026 Jan 13. doi:10.1097/CCM.0000000000007016.
13. Zhu M, Zhang Y, Liu W. Vasoactive agent therapy in septic shock: from monotherapy battles to tailored hemodynamic optimization. Shock. 2025 Dec 23. doi:10.1097/SHK.0000000000002781.
14. Kravitz MS, Kattouf N, Stewart IJ, Ginde AA, Schmidt EP, Shapiro NI. Plasma for prevention and treatment of glycocalyx degradation in trauma and sepsis. Crit Care. 2024;28(1):254. doi:10.1186/s13054-024-05026-7.
15. Kelly EJ, Ziedins EE, Carney BC, Moffatt LT, Shupp JW. Endothelial dysfunction is dampened by early administration of fresh frozen plasma in a rodent burn shock model. J Trauma Acute Care Surg. 2024;97(4):520-528. doi:10.1097/TA.0000000000004373.
16. Scheck M, Velten M, Klaschik S, Soehle M, Frede S, Gehlen J, et al. Differential modulation of endothelial cell function by fresh frozen plasma. Life Sci. 2020;254:117780. doi:10.1016/j.lfs.2020.117780.
17. Dietrich M, Hölle T, Lalev LD, Loos M, Schmitt FCF, Fiedler MO, et al. Plasma transfusion in septic shock—A secondary analysis of a retrospective single-center cohort. J Clin Med. 2022;11(15):4367. doi:10.3390/jcm11154367.
18. Qin X, Zhang W, Zhu X, Hu X, Zhou W. Early fresh frozen plasma transfusion: is it associated with improved outcomes of patients with sepsis? Front Med (Lausanne). 2021;8:754859. doi:10.3389/fmed.2021.754859.
19. Brandtner AK, Lehner GF, Pircher A, Feistritzer C, Joannidis M. Differential procoagulatory response of microvascular, arterial and venous endothelial cells upon inflammation in vitro. Thromb Res. 2021;205:70-80. doi:10.1016/j.thromres.2021.07.002.
20. Torres Filho IP, Torres LN, Salgado C, Dubick MA. Plasma syndecan-1 and heparan sulfate correlate with microvascular glycocalyx degradation in hemorrhaged rats after different resuscitation fluids. Am J Physiol Heart Circ Physiol. 2016;310(11):H1468-H1475. doi:10.1152/ajpheart.00006.2016.
21. Dietrich M, Hölle T, Lalev LD, Loos M, Schmitt FCF, Fiedler MO, et al. Plasma transfusion in septic shock—A secondary analysis of a retrospective single-center cohort. J Clin Med. 2022;11(15):4367. doi:10.3390/jcm11154367. (Nota: duplicada con la 17; conviene eliminar una para evitar repetición.)
22. Qin X, Zhang W, Zhu X, Hu X, Zhou W. Early fresh frozen plasma transfusion: is it associated with improved outcomes of patients with sepsis? Front Med (Lausanne). 2021;8:754859. doi:10.3389/fmed.2021.754859. (Nota: duplicada con la 18; conviene eliminar una.)
23. Clausen NE, Meyhoff CS, Henriksen HH, et al. Plasma as endothelial rescue in septic shock: a randomized, phase 2a pilot trial. Transfusion. 2024;64(9):1653-1661. doi:10.1111/trf.17939.
24. Sendor R, et al. Confounding by indication and the role of active comparators. Pharmacoepidemiol Drug Saf. 2022. doi:10.1002/pds.5407.
25. McMahon AD. Approaches to combat confounding by indication in observational studies of intended drug effects. Pharmacoepidemiol Drug Saf. 2003;12(7):551-558. doi:10.1002/pds.883.
26. White SK, Walker BS, Schmidt RL, Metcalf RA. The incidence of transfusion-related acute lung injury using active surveillance: a systematic review and meta-analysis. Transfusion. 2024;64(2):289-300. doi:10.1111/trf.17688.
27. White SK, Walker BS, Potter S, Anderson D, Metcalf RA. Estimating the incidence of transfusion-associated circulatory overload using active surveillance: a systematic review and meta-analysis. Transfusion. 2025;65(6):1061-1071. doi:10.1111/trf.18258.
28. AABB. Transfusion-associated circulatory overload (TACO): 2018 surveillance definition. Bethesda (MD): AABB; 2018.
29. von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies. PLoS Med. 2007;4(10):e296. doi:10.1371/journal.pmed.0040296.
Received: December 3, 2025 / Accepted: February 21, 2026 / Published (online): March 15, 2026 (Europe/Madrid)
Citation. Vélez-Páez JL, Ochoa-Robles E, Correa F, Jiménez G. Personalized treatment with Fresh Frozen Plasma for the pro-inflammatory septic phenotype characterized by refractory shock, hemodynamic incoherence, and endothelial damage. A proof of concept. BioNatura Journal: Ibero-American Journal of Biotechnology and Life Sciences. 2026;3(1):10. https://doi.org/10.70099/BJ/2026.03.01.10
Correspondence should be addressed to: jlvelez@uce.edu.ec
Peer Review Information BioNatura Journal thanks the anonymous reviewers for their valuable contribution to the peer-review process. Regional peer-review coordination was conducted under the BioNatura Institutional Publishing Consortium (BIPC), involving:
• Universidad Nacional Autónoma de Honduras (UNAH)
• Universidad de Panamá (UP)
• RELATIC (Panama)
Reviewer selection and assignment were supported via: https://reviewerlocator.webofscience.com/
• Universidad Nacional Autónoma de Honduras (UNAH)
• Universidad de Panamá (UP)
• RELATIC (Panama)
Reviewer selection and assignment were supported via: https://reviewerlocator.webofscience.com/
Publisher Information Published by Clinical Biotec S.L. (Madrid, Spain) as the publisher of record under the BioNatura Institutional Publishing Consortium (BIPC). Places of publication: Madrid (Spain); Tegucigalpa (Honduras); Panama City (Panama). Online ISSN: 3020-7886.
Open Access Statement All articles published in BioNatura Journal are freely and permanently available online immediately upon publication, without subscription charges or registration barriers.
Publisher’s Note BioNatura Journal remains neutral regarding jurisdictional claims in published maps and institutional affiliations.
Copyright and License © 2026 by the authors. This article is published under the terms of the Creative Commons Attribution (CC BY 4.0) license, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
License details: https://creativecommons.org/licenses/by/4.0/
License details: https://creativecommons.org/licenses/by/4.0/
Governance For editorial governance and co-publisher responsibilities, see the BIPC Governance Framework (PDF) at: https://clinicalbiotec.com/bipc
