AI as the Catalyst for a
New Paradigm in Biomedical Research
ABSTRACT
This editorial
examines how artificial intelligence (AI)—including machine learning,
generative AI, and natural language processing—is reshaping biomedical research
and pharmaceutical R&D. It outlines distinct adoption archetypes emerging
among large pharmaceutical organizations: partnership-driven acceleration
through strategic technology alliances; culture-centric transformation that
embeds AI into everyday scientific and operational decision-making; and
production-first democratization that makes AI tools broadly usable across
functions. In parallel, AI is lowering entry barriers for smaller biotech
companies, enabling faster iteration in molecular design and earlier clinical
translation, while cloud and federated approaches expand access to powerful pre-trained
models without compromising proprietary data. The editorial also emphasizes the
limiting factors that will determine whether "democratized discovery"
translates into sustained impact: high-quality, interoperable data; rigorous
model validation; transparency and auditability; workforce upskilling; ethical
oversight; and alignment with evolving regulatory expectations. Together, these
elements define a pragmatic pathway toward an AI-integrated biomedical
ecosystem focused on speed, safety, and equitable innovation.
Keywords:
Artificial intelligence, pharmaceuticals, biomedicine, innovation, ethics,
regulation, technological alliances, drug discovery, startups, democratization.
Editorial
The shift toward an
AI-integrated pharmaceutical ecosystem is no longer a peripheral strategy but a
central imperative for global health innovation. The landscape of biomedical
and scientific research is undergoing a fundamental transformation. While the
impact of artificial intelligence (AI) may not be immediate, the current
adoption of machine learning (ML), generative AI (GenAI), and natural language
processing (NLP) across the value chains of major pharmaceutical entities
signals a permanent shift away from traditional R&D methodologies. This
transition is not simply technological but a reconfiguration of institutional
agility and scientific reach1, as evidenced by the rapid development
of mRNA platforms during recent worldwide health crises .
Strategic Divergence in AI
Adoption
Analysis of industry
leaders discloses clear archetypes of AI integration, delivering a roadmap for
organizational evolution. These strategies demonstrate how AI is being utilized
to overcome the traditional "Eroom's Law" (the slowing of R&D
productivity) by boosting predictive accuracy and operational speed 2,3.
- The Partnership-Driven Efficiency Model (Pfizer): This model focuses on a combination of internal R&D and targeted external collaborations with technology giants such as NVIDIA and AWS. Using these partnerships, the organization has successfully compressed drug development timelines—most notably for Paxlovid—from years to just 30 days in phase 3 research.
·
The
Culture-Centric Transformation Model (Moderna):
Positions AI as a core business function led by executive vision. By deploying
over 3,000 customized GPTs and merging HR with IT, the organization builds a
"human-AI workforce." This approach allows for the scaling of complex
mRNA sequences and manufacturing protocols with a significantly leaner team
than traditional pharma giants 4.
·
The
Democratized Production-First Model (Sanofi): Aims
to be the first pharma company powered by AI at scale by making tools
"snackable" and accessible to all employees. Platforms like the Aily
app and Plai empower real-time decision intelligence across the supply
chain and R&D, leveraging specialized models such as CodonBERT for
protein engineering 5.
The Democratization of
Discovery
AI is significantly
leveling the playing field for small emerging biotech firms. Generative AI
platforms have already enabled the first fully AI-discovered molecules to enter
Phase 2 clinical trials, specifically targeting conditions such as idiopathic pulmonary
fibrosis 6. This opening up allows startups to apply tools such as DragonFold
for atomic-resolution protein design and cloud-based federated platforms such
as TuneLab to access powerful, pre-trained models while maintaining the
privacy of their proprietary data. These advances in protein structure
prediction, driven by breakthroughs such as AlphaFold, have fundamentally
changed the starting point for drug discovery 7.
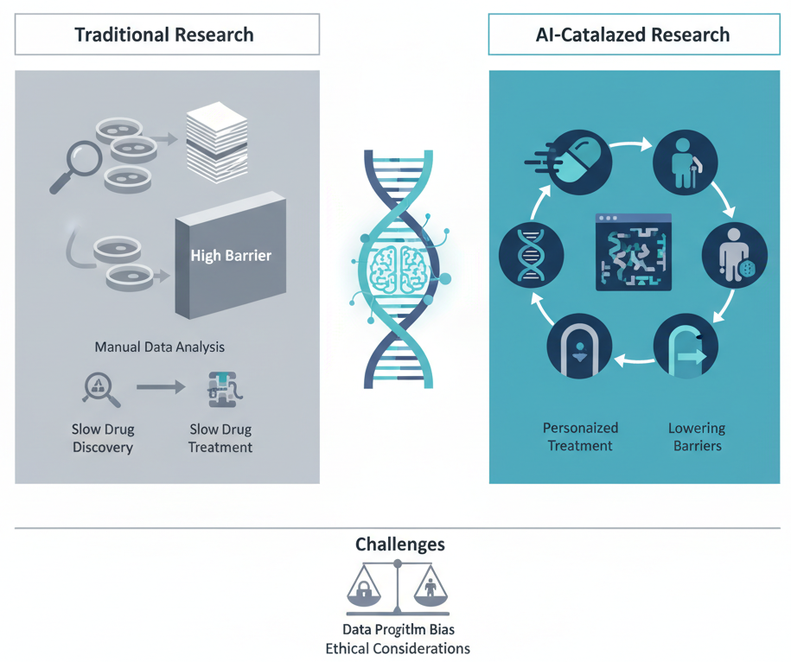
Figure
1. Transformation of the Biomedical R&D Model. Comparison between
traditional research—centered on manual data analysis, higher entry barriers,
and slower discovery/treatment cycles—and an AI-catalyzed research framework
oriented to agility and personalized treatment. The diagram summarizes the shift
toward “democratized discovery” while highlighting key implementation
challenges, including data/algorithmic bias and ethical oversight, alongside
broader requirements for interoperability and regulatory governance. AI
transparency note: The graphic layout and icon arrangement were assisted
using GPAI (gpai.app); the conceptual design, scientific
content, and final wording were defined, reviewed, and validated by the authors.
Handling the Challenges
Ahead
Despite these
advancements, the path to full AI maturity faces substantial structural
hurdles. Organizations must manage the interoperability of diverse internal and
external platforms to guarantee data consistency. Furthermore, shifting toward
a human-AI workforce requires extensive employee education and a culture of
"responsible AI" to ensure moral oversight. Finally, as AI begins to
predict regulatory queries and optimize submissions, preserving transparency
and compliance with evolving global standards remains a continual challenge for
the industry.
CONCLUSION
The transition
toward AI-enabled biomedical research is best understood as an institutional
transformation rather than a purely technological upgrade. Competitive
advantage will come not only from better algorithms, but from disciplined
execution: interoperable data foundations, fit-for-purpose validation, and
transparent governance that keeps responsibility and accountability clearly
human. Organizations that treat AI as a workforce capability—supported by
training, incentives, and cross-functional integration—will move from isolated
pilots to scalable, reproducible impact.
At the same
time, the promise of "democratized discovery" will be realized only
if high standards for quality, ethics, and regulatory readiness match access to
tools. As AI increasingly influences experimental prioritization, development
decisions, and regulatory interactions, the sector must preserve trust through
auditability, bias mitigation, and clear documentation of model intent, limits,
and uncertainty. In this context, progress is measured not by speed alone, but
by reliable translation into safer, more effective, and more accessible health
solutions.
Funding
This research received no external funding.
This research received no external funding.
Acknowledgments
The author would like to thank the BioNatura Journal editorial board for their support. No specific grants from public, commercial, or not-for-profit funding agencies were used for this work.
The author would like to thank the BioNatura Journal editorial board for their support. No specific grants from public, commercial, or not-for-profit funding agencies were used for this work.
Conflicts of Interest
The author declares no conflict of interest.
The author declares no conflict of interest.
Artificial Intelligence (AI) Use Declaration
Generative AI tools
were used only for language/format editing and to assist in drafting Figure
1 using GPAI (gpai.app), under full human
supervision. No AI was used for data generation/analysis/interpretation.
Authors reviewed and verified all content and the final figure per BioNatura
Journal AI policy (https://bionaturajournal.com/artificial-intelligence--ai-.html).
REFERENCES
1. Baden LR, Pajon R, et al. Efficacy and Safety of
the mRNA-1273 SARS-CoV-2 Vaccine. N Engl J Med. 2021;384(5):403-416.
doi:10.1056/NEJMoa2035389.
2. Vamathevan
J, et al. Applications of machine learning in drug discovery and development.
Nat Rev Drug Discov. 2019;18(6):463-477. doi:10.1038/s41573-019-0024-5.
3. El-Sayed
SM, et al. Artificial intelligence in drug discovery and development. Drug
Discov Today. 2023;28(3):103483. doi:10.1016/j.drudis.2023.103483.
4. OpenAI.
Moderna and OpenAI: Accelerating the development of life-saving treatments
[Case Study]. 2024. Available from: https://openai.com/index/moderna/
5. Sanofi
S.A. Sanofi aims to become first pharma company powered by artificial
intelligence at scale [Press Release]. 2023 Jun 13.
6. Ren F, et
al. A small-molecule TNIK inhibitor targets fibrosis in preclinical and
clinical models. Nat Biotechnol. 2024;42:1-13. doi:10.1038/s41587-024-02143-0.
7. Jumper J,
et al. Highly accurate protein structure prediction with AlphaFold. Nature.
2021;596(7873):583-589. doi:10.1038/s41586-021-03819-2.
Received: Jan 10 2026 / Accepted: Feb 21 2025 / Published (online): Mar 15 2026
(Europe/Madrid)
Citation. Pajon R. AI
as the Catalyst for a New Paradigm in Biomedical Research. BioNatura
Journal: Ibero-American Journal of Biotechnology and Life Sciences.
2026;3(1):1. https://doi.org/10.70099/BJ/2026.03.01.1
Additional Information
Correspondence should be addressed to: Email: Rolando Pajon: rolando.pajon@biofabri.es
Peer Review Information
The BioNatura Journal
Editorial Office internally reviewed this Editorial in accordance with the
journal's policies for editorials, perspectives, and commentaries.
BioNatura Journal thanks the scientific advisors who contributed expert feedback during the editorial preparation process.
Regional editorial coordination was conducted under the BioNatura Institutional Publishing Consortium (BIPC), involving Universidad Nacional Autónoma de Honduras (UNAH), Universidad de Panamá (UP), and RELATIC (Panama).
BioNatura Journal thanks the scientific advisors who contributed expert feedback during the editorial preparation process.
Regional editorial coordination was conducted under the BioNatura Institutional Publishing Consortium (BIPC), involving Universidad Nacional Autónoma de Honduras (UNAH), Universidad de Panamá (UP), and RELATIC (Panama).
Publisher Information
Published by Clinical Biotec S.L.
(Madrid, Spain) as the publisher of record under the BioNatura Institutional
Publishing Consortium (BIPC).
Institutional co-publishers: Universidad Nacional Autónoma de Honduras (UNAH), Universidad de Panamá (UP), and RELATIC (Panama).
Places of publication: Madrid (Spain); Tegucigalpa (Honduras); Panama City (Panama).
Online ISSN: 3020-7886
Institutional co-publishers: Universidad Nacional Autónoma de Honduras (UNAH), Universidad de Panamá (UP), and RELATIC (Panama).
Places of publication: Madrid (Spain); Tegucigalpa (Honduras); Panama City (Panama).
Online ISSN: 3020-7886
Open Access Statement
All articles published
in BioNatura Journal are immediately and permanently available online in
open-access format, without subscription or registration barriers.
Publisher's Note
BioNatura Journal
remains neutral with regard to jurisdictional claims in published maps and
institutional affiliations.
Copyright and License
© 2026 by the authors.
This Editorial is published under the terms of the Creative Commons
Attribution (CC BY 4.0) license, which permits unrestricted use, distribution, and
reproduction in any medium, provided the original work is properly cited.
License details: https://creativecommons.org/licenses/by/4.0/
License details: https://creativecommons.org/licenses/by/4.0/
Governance
For full editorial
governance, co-publisher responsibilities, and consortium structure, see the BIPC Governance
Framework (PDF) available at:
https://clinicalbiotec.com/bipc
https://clinicalbiotec.com/bipc
